Compiled: 07-04-25 | Written by © Alexandra Chambers
Email: Neurotopialincoln@outlook.com
1: Executive Summary
This investigative report uncovers global systemic corruption, regulatory capture, and the deliberate manipulation of scientific narratives within multiple systems including the pharmaceutical industry over the past few decades. Through extensive analysis, it exposes how corporate profit motives have repeatedly taken precedence over public health, leading to harmful policies and medical interventions.
Focusing on divalproex sodium (Depakote), sodium valproate (Epilim/Epilim Chrono), and pteroylmonoglutamic Acid (commonly known as synthetic folic acid), this report examines the sequence of events leading to the widespread fortification of food and the consequences of these interventions. When tracing the historical context and regulatory decisions surrounding these drugs, a consistent pattern emerges -one where industry-driven narratives have shaped public policy at the expense of long-term health outcomes.
This report will dissect:
- The pharmaceutical industry’s role in shaping scientific consensus through selective data publication, suppression of adverse findings, and financial influence over regulators.
- The historical precedent of pharmaceutical negligence, from thalidomide to valproate, highlighting recurring patterns of delayed safety recognition and harm minimization tactics.
- The hidden risks of synthetic folic acid fortification, including its biochemical instability, links to increased disease risk, and the overlooked impact on populations with genetic vulnerabilities.
Through rigorous analysis, this report challenges the prevailing narratives, demands accountability, and encourages a re-evaluation of policies that prioritize corporate interests over public well-being.
2: Historical Context: Thalidomide, Valproate, and Global Corruption
Pharmaceutical negligence has repeatedly harmed global populations, from the devastating consequences of Thalidomide (1950s-1960s) to the widespread birth defects caused by Valproate (Depakote/Epilim). Early recognition of thalidomide’s teratogenic effects were documented in The Lancet, where Somers (1962) discussed the emerging link between prenatal exposure and congenital anomalies. Despite early warnings, industry-driven delays in regulatory action allowed these drugs to cause catastrophic harm on a global scale. The thalidomide tragedy alone resulted in over 10,000 severe birth defects worldwide yet regulatory intervention was delayed until public outcry forced action (Somers, 1962).
The Illusion of Protection: Thalidomide, Paramethadione, and the Strategic Separation of Harm
In mainstream pharmaceutical history, the United States is often portrayed as having narrowly avoided the thalidomide disaster due to the vigilance of one FDA reviewer: Dr. Frances Oldham Kelsey. In 1960, Dr. Kelsey famously refused to approve thalidomide for use in the U.S., citing inadequate safety data -particularly concerns around neurological effects (FDA, 2010).
Dr Kelsey’s actions likely prevented thousands of birth defects and saved lives, earning her widespread recognition. However, the emphasis on this singular act of integrity has had the effect of creating a false sense of protection -an illusion that the U.S. escaped the teratogenic harms other countries suffered.
Trimethadione to Paramethadione
Trimethadione, approved in 1946, was already linked to congenital anomalies. Rather than withdraw it and acknowledge its risks, Abbott Laboratories introduced paramethadione in 1949 -a sister compound, with only a single chemical substitution (methyl to ethyl group). This change did not eliminate teratogenicity, but it did allow them to:
- Patent a “new” product – Paradione.
- Avoid scrutiny under the same data.
- Continue profiting while sidestepping accountability.
This worked -for nearly 45 years.
Paramethadione: The Overlooked Domestic Teratogen
In 1949, the FDA had already approved a different drug: paramethadione, a structural analogue of trimethadione, both part of the oxazolidinedione class of anticonvulsants. Marketed under the name Paradione by Abbott Laboratories, paramethadione was prescribed in the U.S. for absence seizures (NCATS).
By the 1970s, evidence mounted that paramethadione caused congenital malformations, including craniofacial anomalies, growth restriction, cardiac defects, and neurodevelopmental disorders -echoing the harms later seen with thalidomide (German et al., 1970).
Despite this, paramethadione remained available in the United States for decades -without fanfare, moral scrutiny, or public outcry.
Paramethadione (marketed as Paradione by Abbott Laboratories) was distributed in the United States and Canada, where it was prescribed for absence seizures throughout the 1950s and 1960s (CMAJ, 1959; PDR, 1954). To date, aside from Canada, no verified evidence of its approval or distribution has been found in thalidomide-affected regions such as the UK, Australia, or Germany, supporting the theory that paramethadione did not appear in those markets.
This lack of overlap allowed each country’s congenital anomaly data to appear isolated, weakening the global teratogen alert. There was no baseline to compare, no unified call for accountability and the damage was partitioned.
This strategy of pharmaceutical compartmentalization appears to have fragmented the truth.
Dr. Kelsey did act with integrity and likely prevented an immediate public health tragedy, but the narrative constructed around her success has been used -whether intentionally or not -to mask the broader reality:
- The U.S. did not avoid teratogen-induced birth defects.
- It simply experienced them through a different molecule.
- While thalidomide was blocked, paramethadione was already active.
This shifts the focus from a singular act of heroism to systemic orchestration -where one drug is denied to preserve credibility, while another is deployed quietly under the radar.
The case of paramethadione is not an isolated anomaly. It follows a clear, repeating strategy within the pharmaceutical industry: take a drug known or suspected to cause harm, alter it slightly, rename it, and reintroduce it -as if the danger has been resolved.
WHO Pharmacovigilance Systems
While the World Health Organization’s Programme for International Drug Monitoring was established in 1968 as a response to the thalidomide tragedy, its implementation over subsequent decades has raised significant concerns regarding efficacy, transparency, and responsiveness. The system, coordinated through the Uppsala Monitoring Centre, collects adverse drug reaction reports from member states into the VigiBase database (World Health Organization, 2002). However, the programme has been criticized for its reactive model, which relies on post-market surveillance rather than proactive safety mechanisms. As a result, early warning signals are often buried in vast, unfiltered datasets, with limited accountability for action (Lexchin, 2015). Moreover, populations with genetic vulnerabilities – such as individuals with MTHFR mutations, neurodevelopmental conditions, or other atypical metabolic profiles – are frequently excluded from both clinical trials and pharmacovigilance models. This structural oversight allows for the accumulation of long-term, low-grade toxicity that is statistically diluted in broader datasets, making it invisible to the very systems designed to detect harm (Healy, 2012). In this context, the WHO programme, while symbolically important, appears ill-equipped to address the nuanced and intergenerational risks posed by synthetic interventions like folic acid fortification and anti-epileptic drug exposure. Effective pharmacovigilance must go beyond data collection. It must be willing to challenge entrenched regulatory alliances, act on early signals, and include vulnerable subpopulations in its core risk assessments. Without this, the system remains more performative than protective.
2.1 The Valproate Scandal: A Repeating Pattern of Negligence
Epilim (sodium valproate) was introduced in the UK in 1973 (IMMDSR, 2020), supplied by Sanofi. Internal investigations later revealed that Sanofi was aware of its teratogenic effects, yet patients were not given informed consent regarding its risks (The Independent Fetal Anticonvulsant Trust, 2020).
Valproate is highly teratogenic due to its interference with folate metabolism, which is essential for DNA synthesis, repair and methylation (Wei et al., 2008). Folate depletion caused by valproate is particularly dangerous during early pregnancy, when folate is critical for neural tube closure (Nakamura et al., 2020). This is why informed consent is so important and why this kind of drug is unsuitable for women who may become pregnant.
Mechanisms of Folate Depletion:
Inhibition of Folate-Dependent Enzymes (Wei et al., 2008; Reynolds et al., 2022):
- Valproate has been shown to inhibit enzymes in the folate cycle, such as methylenetetrahydrofolate reductase (MTHFR) and dihydrofolate reductase (DHFR). This inhibition can reduce the availability of 5-methyltetrahydrofolate (5-MTHF), the bioactive form of folate required for homocysteine remethylating and DNA methylation.
Impaired Folate Transport (Reynolds et al., 2022):
- Valproate may interfere with folate transport mechanisms, including reduced expression or function of folate transport proteins across the placenta and into neural tissues. This limits fetal folate access during critical windows of development.
Induced Oxidative Stress (Reynolds et al.,2022):
- Valproate can increase oxidative stress and mitochondrial dysfunction, which in turn can increase folate demand. Folate is a key cofactor in oxidative defence, and increased depletion under stress conditions can lead to systemic deficiency.
Increased Folate Utilization (Reynolds et al., 2022)
- By increasing DNA turnover and disrupting mitochondrial function, valproate indirectly increases the body’s need for folate, which may lead to faster depletion of available stores.
Histone Deacetylase (HDAC) Inhibition (Katsuki et al., 2021):
- Valproate is also an HDAC inhibitor. This epigenetic effect can alter gene expression, including genes involved in folate and methylation pathways. These epigenetic disruptions may contribute to abnormal development and long-term neurodevelopmental outcomes.
By the early 1990s, multiple studies confirmed a 10-fold increased risk of severe birth defects in valproate-exposed pregnancies (Moore et al., 2000; Adab et al., 2004). Later research showed a correlation with cognitive impairments and lower IQ in children (Meador et al., 2009). Regulatory bodies issued delayed warnings (EMA, 2009), and UK inquiries later confirmed systemic failings (Cumberlege, 2020).
Strategy: Reset the Narrative, Preserve the Market
The two cases trimethadione → paramethadione, and valproate → Depakote—follow the same structural blueprint:
- Phase 1: Market a drug, collect silent damage data.
- Phase 2: Reformulate just enough to differentiate chemically.
- Phase 3: Rename, relaunch, and continue distribution.
- Phase 4: Disown prior harm, delay liability, protect profit.
Depakote (Divalproex Sodium), a slightly modified formulation of valproate, was strategically introduced in 2000 (EMC, 2000) to create a legally distinct product, avoiding immediate association with the growing concerns surrounding valproate (Feldman, 2023; Lexchin, J., 2020). Through altering the compound, Abbott Laboratories ensured continued marketing and regulatory evasion in the UK, despite mounting evidence of harm. This deliberate regulatory inertia also led to data manipulation, distorting public health narratives and influencing medical practice long before formal policy changes were enacted (US Department of Justice, 2012)
For decades, women were prescribed a drug that authorities knew could cause harm. Their children paid the price –many with lifelong disabilities, many without legal recourse (The Independent Fetal Anticonvulsant Trust, 2020; European Medicines Agency, 2009; U.S. Department of Justice, 2012). Behind the quiet reformulation of Depakote was not just a pharmaceutical strategy, but a structural betrayal of medical ethics. This is not simply a regulatory failure; it is a humanitarian one.
2.2 A Pattern That Extends to Folic Acid Fortification
As history repeats itself, the same corporate strategies, regulatory capture, and data suppression tactics that delayed action on thalidomide and valproate are now being deployed to push synthetic folic acid on a global scale. With an abundance of evidence of its long-term health risks, particularly its association with unmetabolised folic acid (UMFA) accumulation, cancer risks, and neurological impact, this report explores whether global fortification policies have been similarly manipulated to conceal potential dangers.
3: Pharmaceutical and Scientific Language Manipulation and Data Misrepresentation
3.1 Data Misrepresentation: (See also The Zig Zag Theory)
The Zigzag Theory refers to a systemic, geo-temporal distortion strategy used to fabricate the appearance of policy success while masking true causality. It involves asynchronous implementation of regulatory actions across regions, silent internal withdrawals, and selective surveillance practices that collectively distort public health data.
Core Mechanisms:
- Asynchronous intervention timing: Regulatory or manufacturing changes occur in one region while being delayed or omitted in another, enabling global data to be skewed in favour of intervention narratives (e.g. fortification or drug approval).
- Silent internal actions: Pharmaceutical companies quietly cease production, rebrand, or alter formulations long before official disclosures, erasing key variables from public health datasets.
- Surveillance lag and publication delay: Policy outcomes are assessed using stale or partial data, which reflects outdated exposures and misattributes effects to new interventions.
- Geographic confounder erasure: Global health data is merged without adjusting for regional differences in exposure, regulation, or diagnosis, allowing one country’s quiet success to bolster another’s ongoing failure.
Examples:
- Valproate: Regulatory action delayed in the UK compared to the U.S. and Canada. Global data pools ignored these disparities, falsely validating folic acid fortification as the driver of NTD reductions (De Wals et al., 2007). In January 2015, the MHRA issued a formal letter to healthcare professionals warning of the 30–40% risk of developmental disorders and approximately 10% risk of congenital malformations from in utero exposure to valproate, urging stricter prescribing controls and enhanced patient counselling (MHRA, 2015).
- Folic acid fortification, mandated in the United States in 1998, had already been implemented voluntarily in the UK years earlier through industry-driven practices. Early declines in neural tube defect (NTD) rates due to stricter teratogen prescribing preceded official mandates, creating a misleading illusion of policy success (Crider, Bailey and Berry, 2011).
Paramethadione: A teratogen quietly removed in Canada after its official last FDA order in 1989, making it impossible to attribute outcome shifts to known regulatory action (FDA, N.D). (See Report Section 12 – Foundational Studies).
- Although the MHRA began warning of valproate’s teratogenic risks as early as 2009, meaningful restrictions and prescribing reforms were delayed until years later (MHRA, 2009; House of Commons Health and Social Care Committee, 2021).
- FDA Delay (2013): Stronger pregnancy risk warnings issued after prescription rates had already fallen. (FDA, 2013; Baker et al., 2018)
- EMA Reclassification (2014): Required additional risk mitigation, but sales data already showed decline. (EMA, 2014; Hirst et al., 2018)
Through delaying official announcements while internally restricting high-risk drugs, regulatory bodies contributed to a misleading correlation between public intervention policies – such as folic acid fortification – and reduced neural tube defect (NTD) rates (Crider, Bailey and Berry, 2011; House of Commons, 2021). This artificial association bolstered narratives supporting synthetic folic acid, despite the fact that much of the decline began prior to formal mandates. Simultaneously, pharmaceutical and public health actors created data distortions, allowing them to claim that NTD prevalence was declining – even as valproate-exposed pregnancies continued to show persistently high rates of defects (MHRA, 2015; Jentink et al., 2010; Martin, 2017).
3.2 Manipulation of Language
Governments and public health organizations have misrepresented natural nutrient deficiencies to strengthen fortification policies.
Key Language Distortions:
- “Folic acid deficiency” (Department of Health and Social Care, 2024): There is no natural dietary requirement for folic acid – only folate.
- Folate vs. folic acid (scientific confusion): Many studies use these terms interchangeably, hiding key biochemical differences (Obeid, R and Hermann, W., 2019).
- “Neural tube defect prevention” (policy rhetoric): Fails to account for genetic variations in folic acid metabolism (Li, Y et al., 2020).
- In a 2021 study on 6-formylpterin, the author(s) included “folate” in the title, potentially misleading readers into believing both forms of Vitamin B9 degrade into the same toxic byproducts (Goossens et al., 2021).
The 2015 World Health Organization (WHO) guideline on folate thresholds for neural tube defect (NTD) prevention made crucial biochemical distinctions between naturally occurring folate and synthetic folic acid. These distinctions were essential for:
- Understanding metabolic differences.
- Avoiding unintended toxicity.
- Designing equitable public health policy.
Yet, from 2023 onward, public-facing communications – especially in the UK – erased this clarity, rebranding all folate deficiency as “folic acid deficiency” and presenting fortification as a universal solution.
- Scientific distinction: WHO (2015).
- Policy simplification: UK Government Press Release (2024).
3.3 Valproate, Epilim and Depakote – a Triad of Confusion
Further compounding this issue is the intentional interchangeability of drug names – Depakote, Epilim, and sodium valproate – across scientific literature and clinical records. Sodium valproate (Depakote) has multiple distinct names for the exact sameactiveexcipient, but a slightly varied formulation. This varied formulation, justified as an adjustment for side effects and prevented clear tracking of prescription rates and teratogenic outcomes of the drug(s).
Key Timeline of Naming Manipulation:
- 1978: Valproate introduced under multiple brand names worldwide (EMA, 2018; FDA, 2023)
- 1990s: Depakote branding becomes dominant in US, while valproate is still used in Europe. (FDA Orange Book, 2023)
- 2000s: Various studies begin to cite either “valproate” or “Depakote” inconsistently, preventing clear tracking of adverse effects. (MHRA 2018, EMA 2014; EMA 2018)
- 2010s-Present: Regulatory warnings often mention only one name at a time, making it difficult to assess full global impact. (MHRA, 2021; FDA, 2023)
Through splitting data tracking between ‘valproate’ and ‘Depakote,’ pharmaceutical companies ensured that full teratogenic impact remained obscured. (Hirst et al., 2018)
This strategy, documented across MHRA, FDA, and EMA reports, represents a systematic effort to control narrative accountability by burying harm within semantic confusion (FDA, 2013; EMA, 2018).
The interchangeable use of drug brand names – Depakote, Epilim, and the generic name sodium valproate – across scientific literature and clinical records has:
- Obstructed efforts to track consistent exposure data (MHRA 2018; MHRA 2021; EMA 2014; House of commons health and social care committee 2021).
- Complicated attribution of birth defects and neurodevelopmental disorders (Christensen et al., 2013).
- Enabled the misattribution of harm prevention to folic acid, rather than to teratogens like valproate (Abbott Laboratories, 2012).
These distortions not only obscure biochemical pathways and genetic vulnerability (MTHFR, etc.) but have also allowed teratogenic risk to persist for decades in clinical and regulatory silence.

3.4 Data Skewing – Interchangeable Use of Folic Acid & Folate in Studies
Many major studies that claim to prove folic acid effectiveness measured folate intake. By switching terminology mid-study or misrepresenting findings in abstracts, public health bodies created a misleading scientific consensus.
Examples of Low – Key Study Manipulation:
- Numerous public health conclusions on neural tube defect prevention have been based on a mixed body of research that often fails to distinguish between naturally occurring folate and synthetic folic acid. This conflation is evident in studies and policy guidance, as noted by Lucock (2000), Smith et al. (2008), and Pfeiffer et al. (2015), despite the substantial metabolic differences between these compounds – especially in genetically vulnerable populations.
“Many studies and policies do not distinguish clearly between folate and folic acid, despite differences in bioavailability and metabolism” – Lucock (2000) – British Journal of Biomedical Science
The CDC’s early evaluation of folic acid fortification outcomes in 1998 relied on dietary intake surveys that also did not distinguish between natural folate and synthetic folic acid. While these findings were used to support the benefits of fortification, they were based on estimated “folate equivalents,” conflating naturally occurring and fortified sources (CDC, 1998). This conflation undermines the precision of conclusions drawn about the specific effects of synthetic folic acid, especially in populations with genetic vulnerabilities to UMFA accumulation. Through artificially boosting folic acid intake, industry-created data distortions allowed them to claim that NTD prevalence was declining, even when valproate-exposed pregnancies showed persistently high rates of defects.
4: Abbott, AbbVie and Pharmaceutical Regulatory Capture
Abbott, AbbVie, and Pharmaceutical Regulatory Capture
Abbott Laboratories, a multinational pharmaceutical and healthcare corporation, has a long-standing history of prioritizing profit over public safety. This pattern has repeatedly endangered some of the most vulnerable populations – pregnant women, unborn children, and premature infants – through its aggressive marketing of high-risk products and failure to communicate known dangers.
One of Abbott’s most notorious products, Depakote (divalproex sodium), an anticonvulsant and mood stabilizer prescribed for epilepsy, bipolar disorder, and migraines, was aggressively marketed to women of childbearing age despite well-documented teratogenic risks. These included:
- Neural tube defects (NTDs): Depakote is strongly linked to spina bifida, anencephaly, and other major NTDs (Rosa, 1991; Jentink et al. 2010).
- Facial and limb malformations: Thousands of exposed children were born with cleft palates, cardiac anomalies, and malformed limbs (Bouboushian, 2012; Moore et al. 2000).
- Cognitive impairments: Children exposed in utero were found to have significantly lower IQs and a higher likelihood of neurological damage (Meador et al, 2009; Moore et al, 2000).
By the time legal action forced Abbott to acknowledge these harms, countless children had already been born with preventable lifelong disabilities. The company’s failure to secure informed consent from prescribing physicians or pregnant patients underscores the deep ethical violations underpinning its business strategy.
4.1 Corporate Fraud, Legal Repercussions, and the Infant Formula Scandal
In 2012, Abbott Laboratories pleaded guilty to illegally marketing Depakote and agreed to pay a $1.5 billion settlement, one of the largest in pharmaceutical history (U.S. Department of Justice, 2012). The company had promoted the drug for off-label uses – including for elderly dementia patients and unapproved psychiatric conditions – despite known and devastating side effects (United States District Court, Southern District of Illinois, 2017).
Further compounding its record of negligence, Abbott has also come under fire for its infant formula products. In 2024, the company was ordered to pay $495 million to settle claims linking its cow’s milk-based formula to necrotizing enterocolitis (NEC) in premature infants – a severe gastrointestinal condition that can lead to death or long-term disability. Investigations revealed that internal documents had flagged the risk, but Abbott failed to adequately warn healthcare providers or caregivers (Burnson & Feeley, 2024; Bloomberg Law, 2024).
This expanding litigation portfolio reflects Abbott’s systemic approach: selling high-risk products without transparency, minimizing liability through delayed admissions, and only addressing public health consequences when forced by legal settlements. In 2013, Abbott strategically spun off its branded pharmaceutical division as AbbVie, distancing itself from regulatory scrutiny and shielding its assets from lawsuits stemming from past misconduct (SEC Filings, 2013; AbbVie, 2013).
Abbott’s actions exemplify regulatory capture – where powerful pharmaceutical entities influence or evade oversight structures meant to protect public health.
4.2 AbbVie: The Spin-Off That Profits from Its Own Damage
AbbVie profits from treating neurological ‘disorders’ (or neurological damage) caused by prenatal valproate exposure). The same drug they made – the one that caused neural tube defects – they are now profiting from. Make the disease > make the cure > reap the profits. A self-sustaining business model, like a snake eating its own tail.
Abbott and AbbVie exemplify the worst of pharmaceutical corruption; hiding dangers, harming the most vulnerable, and profiting off both the cause and the cure. Their tactics have been painstakingly exposed, yet they continue to shape policies, evade full accountability, and expand their market dominance.
- AbbVie invested over $63 million lobbying legislators, ensuring drug safety policies remained favorable to its business interests (OpenSecrets, 2024).
- Deliberate delays in warnings: The FDA rejected and postponed updating pregnancy warnings in 2005 and 2007, after closed-door discussions with Abbott (FDA, 2007; Rheinfrank v. Abbott Laboratories, 2016).
- Industry influence over public health: This is a textbook example of ‘regulatory capture’, where industry lobbying distorts public health policy to protect corporate profits (Lexchin, 2005; Light et al., 2013)
4.3 Regulatory Delay Through Suppression of Warnings:
Despite growing evidence of valproate’s teratogenicity and developmental risks, critical updates to pregnancy safety warnings were actively stalled by the U.S. Food and Drug Administration (FDA) following private communications with Abbott Laboratories. In 2005, Abbott submitted a Prior Approval Supplement (PAS) to amend Depakote’s label, warning of potential developmental delay in exposed infants. The FDA rejected the proposal in 2006, stating that the available evidence did not warrant a label change (FDA, 2007; Rheinfrank v. Abbott Laboratories, 2016). A second attempt in 2007 to update the pregnancy warning was similarly denied, despite increasing concern from medical professionals and growing evidence of neurodevelopmental harm.
These regulatory rejections, which remained outside public scrutiny at the time, were later cited in court as “clear evidence” that the FDA would not have permitted Abbott to unilaterally change the label through a ‘change being effected’ (CBE) pathway – thus blocking urgent safety communication that could have protected patients. The delayed recognition of harm allowed Abbott (and later AbbVie) to continue marketing Depakote without meaningful foetal risk disclosure until 2011, when formal updates were finally sanctioned. This timeline reveals a profound failure in pharmacovigilance, highlighting the extent to which industry and regulator collusion can obscure risk, delay justice, and perpetuate harm.

4.4 Deliberate Defect – How Regulatory Delay Manufactured a Crisis to Justify Policy
In the early 2000s, a quietly unfolding tragedy was allowed to persist behind closed doors. Despite mounting evidence linking valproate (Depakote) exposure in utero to neural tube defects (NTDs) and severe neurodevelopmental harm, the U.S. Food and Drug Administration (FDA) repeatedly rejected attempts by Abbott Laboratories to update the drug’s pregnancy safety warnings. In 2005 and again in 2007, Abbott submitted formal Prior Approval Supplements (PAS) requesting label changes to reflect the growing scientific concern. The FDA refused, citing “insufficient evidence” to warrant revised language (FDA, 2007; Rheinfrank v. Abbott Laboratories, 2016).
The rejection of clearer warnings not only ensured that thousands of pregnancies remained exposed to a known teratogen – it also generated the very NTD outcomes that could later be used to justify mandatory synthetic folic acid fortification. In essence, the regulators allowed a preventable wave of birth defects to continue so that the subsequent decline – falsely attributed to folic acid – could be politically and scientifically leveraged.
When the time warnings were finally updated in 2011, the damage had already been done. NTDs had peaked, the fortification narrative had been embedded into global health policy, and the pharmaceutical industry had successfully redirected attention away from drug-induced harm. This orchestrated delay provided a false causal framework: a misleading appearance that folic acid fortification was responsible for declining NTD rates, when in reality the decline correlated more closely with restricted use of high-risk medications like valproate and Depakote.
This manoeuvre does not seem accidental – it appears to be strategic regulatory choreography – where human harm became a statistical necessity to sell a policy, to protect a product, and to preserve an illusion of control. Delayed warnings became a tool of both plausible deniability and manufactured consent.
To this day, there is no public record of disciplinary action, no apology, and no systemic correction of the error. The regulatory apparatus remains intact, strengthened by the very silence it cultivated.
To understand why such orchestration would occur, one must consider the regulatory and political climate of the late 20th century. Pharmaceutical companies like Abbott, facing increasing scrutiny over teratogenic risks from drugs such as valproate, had every incentive to redirect attention. By inflating neural tube defect (NTD) statistics – delaying safety actions on valproate while allowing widespread exposure – they manufactured a public health crisis that conveniently created demand for a policy intervention already in the pipeline: folic acid fortification.
This manoeuvre served several strategic purposes:
- Deflection of liability: Attributing NTDs to folate deficiency rather than drug toxicity, enabled blame to be shifted away from industry.
- Policy manipulation: Fortification programs, which benefited synthetic folic acid manufacturers, could be rapidly implemented under the guise of public health emergency.
- Patent strategy and revenue continuity: As teratogenicity threatened Depakote’s reputation, these orchestrated events provided a controlled exit strategy and justification for product evolution and evergreening tactics.
In short, the rise in valproate-linked birth defects was not just ignored – it was used – to catalyse a new era of interventionist public health policy that offered immense pharmaceutical and political profit, with minimal accountability.
Public health should never be dictated by corporate profits. Yet, these cases reveal a disturbing reality: when industry influence overrides science, safety, and ethics, the consequences are borne by the innocent – children, families, and the most vulnerable in society.
4.5 CASE SPOTLIGHT: Rheinfrank v. Abbott Laboratories (2016): When truth was proven – but justice denied
In the aforementioned legal case concerning Depakote, Rheinfrank v. Abbott Laboratories (2016), a family sued after their child was born with severe congenital defects caused by prenatal valproate exposure. The case unearthed an important fact: Abbott had submitted formal requests to the FDA in 2005 and 2007 to update Depakote’s pregnancy warnings, citing evidence of developmental harm; the FDA rejected both.
Despite this – despite Abbott’s attempts to warn, and despite the undisputed harm – the jury ruled in Abbott’s favour.
The court concluded that since the FDA had rejected the proposed label changes, Abbott could not legally be held responsible for failing to warn the public. This legal loophole, known as “pre-emption,” created a devastating precedent: regulatory inaction became legal immunity. The message was clear:
If the FDA says no, no one is liable – even when children are born damaged.
The family lost – the child suffered; the warnings came six years too late.
This case is sadly not an anomaly, it is a template. It reveals a coordinated system in which harm is permitted, warnings are delayed, and regulatory refusal becomes a shield for pharmaceutical power.
Unasked, Unanswered: Why Did the FDA Reject the Warnings?
The most haunting aspect of Rheinfrank v. Abbott Laboratories (2016) is not the verdict – it’s the silence. The court acknowledged that Abbott Laboratories submitted Prior Approval Supplements (PAS) to the FDA in 2005 and again in 2007, requesting to update Depakote’s pregnancy label to reflect growing evidence of developmental risk. The FDA rejected both requests, claiming the data was insufficient (U.S. Court of Appeals for the Sixth Circuit, 2016; FDA, 2013).
This rejection was not born of ignorance; it followed decades of mounting, published warnings. The real question is why the FDA would reject Abbotts label change.
The evidence of risk was not emerging – it was already well established:
- As early as the 1980s, animal studies and early clinical observations linked valproate to neural tube defects (NTDs) like spina bifida (Dansky and Finnell, 1991; Rosa, 1991).
- A landmark 1991 study in The New England Journal of Medicine found a strong association between valproic acid exposure and spina bifida in infants (Lammer et al., 1991).
- By the 1990s, the teratogenicity of valproate was widely acknowledged, with studies citing a 10–20-fold increased risk of congenital malformations compared to baseline (Arpino et al., 2000; Jentink et al., 2010).
Despite this, the FDA waited until 2011 to approve updated warnings about developmental delay and reduced IQ, following further studies confirming neurodevelopmental harm (Meador et al., 2006; Meador et al., 2013).
This is not a case of science catching up – it is a case of regulatory suppression.
The FDA’s refusal to approve label updates in 2005 and 2007 did not reflect a lack of evidence – It reflected a deliberate decision not to act on it.
That decision:
- Prolonged foetal exposure under outdated safety guidance.
- Allowed NTD rates to rise artificially, strengthening the public health justification for mandatory folic acid fortification.
- And ultimately created legal insulation for Abbott, who could not be held liable due to FDA inaction.
This pattern of evidence suggests more than delay. It suggests design; a design where regulatory silence was weaponized to protect policy and profit over human life.
4.6 Political Influence & Systemic Corruption
AbbVie strategically donates to both Democratic and Republican candidates in the U.S., ensuring continued political influence regardless of election outcomes (Open Secrets, 2024). These financial contributions have directly shaped legislation affecting drug pricing, safety regulations, and market competition.
AbbVie has spent over $100 million lobbying legislators and regulatory agencies, often in opposition to stricter drug safety frameworks that would have affected pregnancy warning updates, labelling transparency, and post-market surveillance. While the company’s lobbying disclosures do not mention valproate by name, these regulatory categories directly govern how drugs like Depakote are controlled and communicated to the public (OpenSecrets, 2024; AbbVie Inc., 2023).
One of the most glaring examples of corporate favoritism is AbbVie’s tax strategy: despite generating over 75% of its revenue from U.S. sales, the company reported only 1% of its revenue for tax purposes in 2020 (US Senate Finance Committee, 2022). This reveals a clear contrast between the tax burdens imposed on ordinary citizens versus corporate giants who exploit legal loopholes to shield their profits.
5: Neural Tube Defect (NTD) Global Birth Trends & Pharmaceutical Influence
This section of the report tracks the historical prevalence of neural tube defects (NTDs) across the UK, US, Italy, Japan, and Australia, cross-referencing these trends with key pharmaceutical events, drug regulations, and folic acid fortification policies. The objective is to identify whether the decline in NTDs is solely due to folic acid fortification, or whether external factors, such as the reduction of teratogenic drugs, played a more significant role.
5.1 Neural Tube Defect (NTD) Data Availability and Limitations by Country/Region
The table below summarizes the most recent and accessible data on NTD prevalence across selected countries, highlighting available raw statistics, source references, and periods where no disaggregated or year-by-year data is publicly available. Absence of such data is treated as a significant limitation in accurate trend comparison for academic and epidemiological analysis.
Upon further analysis, a clear shift in data transparency occurred post 2011. Prior to this, year by year NTD prevalence data was routinely published by EUROCAT and US CDC registries. After this point, most sources transitioned to cumulative summaries or regionally restricted reports, breaking continuity with decades of transparent trend monitoring. This disruption in public data access coincides with the period during which adverse long-term effects of earlier pharmaceutical and policy interventions would be expected to manifest.
| Country | Years Covered | Source | Summary of Available Findings | Missing Data |
| Australia | ~2006–2012 | AIHW | Pre-/post-fortification NTD rate comparison: 14.4% drop | No national annual dataset after 2012 |
| USA | 2016–2020 (estimated) | PMC10898112 (2024), CDC | Estimated prevalence: ~7.0 per 10,000 births; 4,541 spina bifida cases among 3.6M births | No granular national year-by-year data since 2004 |
| England | 2000–2019 (select regions) | PMC10850651 (2024) | Cumulative NTD rates across 5 registers; 1,178 cases among 1.2M births | National year-by-year trends not publicly accessible |
| Scotland | 2000–2021 | BMJ Archives of Disease in Childhood (2025 preprint) | Yearly NTD data (anencephaly, spina bifida, encephalocele) available | None within 2000–2021 anencephaly window |
| France | Not specified | ANSES (2024) | Policy updates on folic acid fortification only | No NTD prevalence data found |
| EUROCAT | 1991–2011 (some regions) | BMJ EUROCAT study | Pooled NTD rates from 28 registries; 9.1 per 10,000 | Post-2011 disaggregated national data inaccessible |
5.2 Global NTD Birth Trends
United Kingdom (UK) NTD Birth Trends:
- 1936–1956: NTD prevalence fluctuated between 1.54 and 2.8 per 1,000 births. (Bower, C. and Stanley, F.J., 2004)
- 1965–1997: NTD prevalence declined by 96%, dropping from 3.80 per 1,000 births in 1965 to 0.14 per 1,000 in 1997. This sharp decline has occurredbefore folic acid fortification is introduced. (McDonnell, R. et al.,1999)
- 1991–2011: Prevalence remained relatively stable, with 2011 rates like 1991. No significant further reduction was observed. (EUROCAT/BMJ, 2015).
- 2011–2021: Recent data indicates that NTD prevalence has slightly increased, contradicting expectations that folic acid fortification would further reduce cases.
- 2010-2014, prevalence was 12.1 per 10,000 total births in Scotland (Stockton, D. et al., 2025).
- 2015-2019, prevalence increased to 13.6 per 10,000 total births. (Best et al., 2024).
- In Scotland (2021), NTD prevalence was 14.3 per 10,000 births, suggesting a continued rise. (Stockton et al., 2025)
NTDs declined significantly before folic acid fortification, largely due to the removal of teratogenic drugs and improved prenatal care. However, recent increases in NTD rates suggest that folic acid fortification has not led to further reductions, raising concerns about its long-term effectiveness.
United States (US) NTD Birth Trends:
A 2016 study released by Stanford Medicine and covered by Erin Digitale, titled “Less decline than expected in rate of brain, spine defects after folic acid fortification program”, claimed that folic acid fortification had reduced neural tube defects (NTDs), but that the decline had since slowed (Digitale, 2016).
While presented as a supportive assessment of fortification policy, the framing of the findings portrays a subtler bias: the study assumes, without fully substantiating, that lower folic acid levels are causatively linked to increased NTD rates – despite existing data suggesting that neural tube defects were already in decline before fortification policies were implemented. This conflation, combined with funding from both the CDC and Stanford’s Department of Paediatrics institutions heavily involved in fortification promotion – raises concerns regarding neutrality.
Rather than investigating alternate explanations for the plateau, such as population-level saturation, adverse effects of excess folic acid, or shifting environmental variables, the study reinforces a one-directional policy narrative. This makes it a powerful example of controlled opposition: appearing to acknowledge flaws in fortification outcomes while still upholding the original policy’s scientific legitimacy and shielding it from deeper scrutiny.
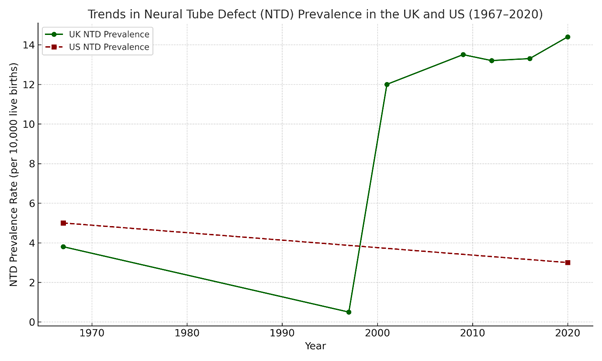
United States (US) NTD Birth Trends: (Centers for Disease Control and Prevention, 2015; CDC, 2023; Mai et al, 2024):
Analysis of publicly available CDC data (2015–2024) reveals a complex trend in neural tube defect (NTD) rates that does not align with claims of uniform decline following folic acid fortification. Instead, recent findings indicate fluctuations in prevalence, raising critical questions about data interpretation and the attribution of causality.
Before folic acid fortification (pre-1998)
- Prior to mandatory folic acid fortification, NTD rates varied significantly by region.
- Estimated spina bifida prevalence was ~5 per 10,000 births in the general population.
After folic acid fortification (1998–Present)
- Following the 1998 fortification mandate, NTD prevalence reportedly dropped by ~28%, with spina bifida rates declining to 3.5 per 10,000 births.
- However, post-2010 data suggests a slowing of decline, with some areas showing plateauing or even slightly rising trends.
The initial reduction in NTDs post-fortification suggests some benefit, but data from 2010 onwards raises questions about whether long-term trends are influenced by fortification alone. Other factors – such as improved prenatal care and reduced use of teratogenic medications – may have played a larger role than acknowledged.
Italy NTD Birth Trends:
Pre-1998 Data (Before Fortification Policies in Other Nations)
- Italy has never implemented mandatory folic acid fortification, making it a useful case study to compare against fortified nations.
- Early studies suggest NTD rates fluctuated but showed gradual declines due to improved medical practices.
Post-2000 Trends
- Italy promotes folic acid supplementation rather than fortification, encouraging women to take folic acid preconceptionally. A Central Italy anomaly study (2002–2004): → 18.5 per 10,000 births in localized area (Stazi et al., 2008)
- Despite no mandatory fortification, Italy has not seen a significantly higher rate of NTDs compared to fortified nations, raising questions about whether fortification is truly necessary for NTD prevention. Folic acid promoted via supplementation guidance, not legislation
(Calzolari et al., 2001) – EU Rare Disease Project
The absence of mandatory fortification in Italy has not resulted in a notable difference in NTD prevalence when compared to fortified countries like the US and Australia. This suggests that other factors – such as dietary folate intake, genetic predispositions, and medical advancements – may be equally or more important.
Australia NTD Birth Trends:
Pre-Fortification Era (Before 2009 – Australian Institute of Health and Welfare, 2014)
- Before mandatory folic acid fortification in wheat flour (2009), Australia’s NTD rate was 4.6 per 10,000 births.
· (Despite voluntary folic acid promotion in 2009, no major decline in NTDs was observed (D’Antoine et al., 2019).
- Following the 2009 fortification mandate, data showed a 14.4% reduction in NTDs per 10,000 conceptions (D’Antoine et al., 2019).
- However, NTD rates remain higher than expected, indicating that fortification did not eliminate the issue as projected.
While folic acid fortification in Australia coincided with a reduction in NTD rates, the decline was not as drastic as predicted. The persistence of NTD cases post-fortification raises questions about whether synthetic folic acid alone is sufficient for prevention – or if biological, environmental, and dietary factors play a larger role.
Japan NTD Birth Trends:
A review of national data in Japan (Nanishi et al., 2019) demonstrates that NTD rates remained relatively stable despite the absence of mandatory folic acid fortification. This data challenges prevailing assumptions about the universality of fortification benefits and highlights the need for region-specific evaluation of public health policies.
Before and After 2000: The Unique Case of Japan
- Japan has never adopted mandatory folic acid fortification, relying instead on natural dietary sources and targeted supplementation.
- Despite this, recent studies suggest that Japan’s total NTD prevalence is not significantly higher than fortified nations – contradicting the claim that fortification is essential.
Recent Data: 2014–2015 Study
- Japan ranked 5th out of 7 developed nations for NTD prevalence, with rates estimated at 8.38–8.74 per 10,000 deliveries.
- However, this was 1.5x higher than previous official estimates, indicating that underreporting may have masked the true prevalence.
Japan’s case is particularly important – it challenges the assumption that fortification is necessary to prevent NTDs. The relatively stable NTD rates despite zero mandatory fortification suggest that alternative strategies, such as dietary folate intake and prenatal care, may be just as effective (or more so) than mass fortification.
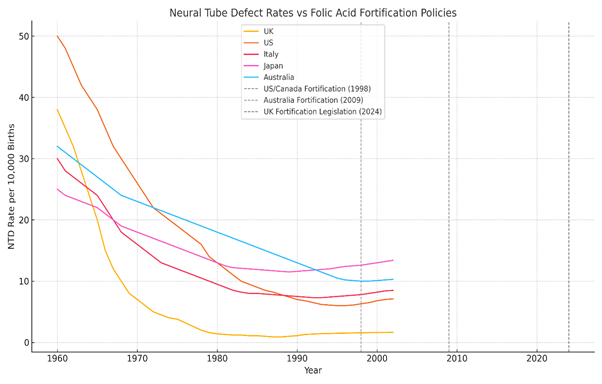
5.3 Global Perspective on Data: Does Fortification Truly Work?
The comparison of these nations reveals an uncomfortable reality:
- Access to raw data has been disrupted since 2011 (Modgil et al., 2022; Wozniak & Bianchi, 2019).
- NTDs declined significantly in many countries before fortification was introduced (De Wals et al., 2007; Wald et al., 2001).
- Countries without fortification (Italy, Japan) have not suffered drastically higher NTD rates and the NTD rates that they do have may well be aligned with their teratogenic drug prescribing practices (D’Angelo et al., 2017; Ueda et al., 2016).
- In fortified nations, NTDs did decline post-fortification, but not to the extent predicted, and it is important to remember that correlation does not equal causation. (Best et al., 2024; Crider et al., 2011).
Recent data suggests that some countries (including the UK and US) may be seeing a plateau and increase in NTD cases, despite ongoing fortification policies (Molley et al., 2023; Greenblatt et al., 2021).
The assumption that mandatory folic acid fortification is the sole reason for NTD reduction is now in serious question. Other factors -including elimination of teratogenic drugs, dietary improvements, genetic factors, and medical interventions – must be considered.
5.4 The Real Reason for NTD Decline: Removing Teratogens, Not Folic Acid?
The mainstream narrative credits folic acid fortification for reducing neural tube defect (NTD) rates – but the data tells a different story. A sharp decline in NTDs occurred well before fortification policies were introduced, suggesting that the true cause of the reduction was the removal of teratogenic (toxic) drugs and environmental exposures.
The Thalidomide disaster (1957–1961)
- One of the most infamous cases of drug-induced birth defects was thalidomide, an anti-nausea drug given to pregnant women in the late 1950s and early 1960s. (McBride, 1961).
- Thalidomide caused severe birth defects, including limb malformations, organ damage, and neural tube defects. (Vargesson, 2009).
- After the drug was banned in the early 1960s, birth defect rates – including NTDs – began to drop dramatically.
Valproate (Epilim/Depakote) and other teratogenic anticonvulsants (1970s–1990s)
- Valproate (sodium valproate, sold as Epilim and Depakote) was widely prescribed for epilepsy, bipolar disorder, and migraines despite early warnings of severe birth defects. (Tomson et al., 2011., UKTIS, 2023).
- Studies confirmed that valproate exposure in pregnancy significantly increased the risk of neural tube defects (including spina bifida) (UKTIS, 2023).
- The drug(s) remained widely used for decades, contributing to persistent NTD cases (MHRA, 2023).
- Only in recent years has the full scale of the “Valproate scandal” been acknowledged, leading to restrictions and lawsuits against manufacturers like Sanofi and Abbott. (House of Commons Health & Social Care Committee, 2020).
- Other anticonvulsants (phenytoin, carbamazepine, phenobarbital) were also linked to NTDs and have been gradually phased out or replaced with safer alternatives for at risk groups. (Meador et al., 2006; Tomson et al., 2011).
The Phasing Out of Leaded Petrol (1970s–1990s)
- Lead exposure is a well-documented neurotoxin that can cause Developmental Disorders, including Neural Tube Defects. (Chen et al., 2022; ATSDR, 2007).
- Leaded petrol (gasoline) was used extensively until the 1980s, exposing pregnant women and fetuses to lead contamination. (UNEP, 2021).
- The gradual removal of leaded petrol in developed countries coincided with a decline in birth defects and neurodevelopmental disorders.
Improved Prenatal Screening and Care (1960s–1990s)
Advancements in prenatal care and maternal health also played a major role in the declining NTD rates:
- Ultrasound technology allowed for early detection of neural tube defects, enabling selective terminations of affected pregnancies if the choice is made, reducing NTD affected births. (Choi et al., 2019; ACOG, 2017).
- Increased awareness of dietary health improved maternal nutrition beyond just folic acid intake. (WHO, 2016).
- Medical guidelines shifted internally away from prescribing high-risk drugs to pregnant or at-risk groups. (UKTIS, 2023).
5.5 Omission by Design – The Untold Impact of Thalidomide on NTDs and the Modern Parallel
While thalidomide’s link to limb deformities has been thoroughly documented, its potential role in neural tube defects (NTDs) has been strategically ignored. Despite a documented 96% decline in NTD prevalence between 1965 and 1997 (McDonnell et al., 1999) – beginning immediately after thalidomide’s withdrawal – there is a conspicuous absence of investigation into this correlation. The regulatory and academic literature focuses almost exclusively on visible limb anomalies, while failing to explore or even hypothesise that the drug’s teratogenic profile may have extended into the neurological domain.
This does not seem to be a gap in evidence; it appears to be a calculated omission.
In avoiding the broader implications of thalidomide’s mechanism – particularly its anti-angiogenic, oxidative, and neural crest cell-disrupting properties (Vargesson, 2009; Kowalski et al., 2016) – the scientific community has evaded accountability for the full scale of foetal harm. The lack of published data does not reflect scientific uncertainty, but rather a wilful narrowing of focus that has effectively erased entire categories of potential damage.
This historical evasion is mirrored today. The same silencing mechanisms that muted thalidomide’s wider teratogenic reach are evident in the current handling of valproate, paramethadione, synthetic folic acid, and emerging mRNA interventions. Regulators continue to approve and promote pharmaceuticals affecting neurodevelopment while downplaying or obscuring evidence of long-term harm – especially in genetically susceptible populations (Bécares et al., 2022; Yadav et al., 2023; Singh et al., 2023).
The pattern is clear:
- A substance causes harm.
- A narrow scope of damage is acknowledged.
- Broader neurological impacts are ignored.
- The affected population bears the burden in silence.
- The cycle repeats.
In refusing to revisit the full teratogenic signature of thalidomide, modern regulators shield themselves from acknowledging that the same systemic failings are being repeated – right now, with different molecules, different branding, and the same devastating silence.
5.6 Did Folic Acid Fortification Really Cause the NTD Decline?
The key question: If NTDs had already declined by over 90% before folic acid fortification, how can policymakers claim that fortification was responsible for the drop?
- In the UK, NTDs dropped from 3.80 per 1,000 births (1965) to 0.14 per 1,000 (1997) – before folic acid fortification was introduced (Wald et al., 2001; Botto et al., 2005).
- In the US, NTDs had been declining for decades before the 1998 fortification mandate (CDC, 1995; Williams et al., 2002).
- In non-fortified countries like Italy and Japan, NTD rates declined without mandatory folic acid (Ueda et al., 2016; D’Angelo et al., 2017).
The data suggests that the elimination of teratogens, improved prenatal healthcare, and better maternal nutrition – not synthetic folic acid – were the real drivers behind the reduction in NTD cases (De Wals et al., 2007; Dolk et al., 2008; Khoshwood et al., 2015), however, the pharmaceutical and food industries have continued to push global flour fortification policies.
In 1996, the U.S. FDA mandated folic acid fortification of enriched grain products, citing public health goals of reducing neural tube defects, and implemented the policy nationwide in 1998 (FDA, 1996).
6: Synthetic Folic Acid: Hidden Toxicity and Health Risks
6.1 Folate vs Folic Acid: What’s the Difference?
While natural folate is essential for health, synthetic folic acid is a structurally distinct compound with different metabolic properties. folic acid, known chemically as Pteroylmonoglutamic Acid (Arnarson, 2024), the synthetic version of vitamin B9, commonly added to fortified foods and supplements. In contrast, folate refers to naturally occurring forms of B9 found in whole foods.
Folate plays a crucial role in:
- Red blood cell formation.
- Supporting foetal development and preventing neural tube defects during pregnancy.
- Enabling DNA/RNA synthesis and repair for healthy cell division (Bailey et al., 2010; Lucock, 2004).
Reference Box: Understanding Folic Acid Levels in Blood
Serum folate (or “folic acid level”) This measures the total folate in the blood – including both natural (active) folate and synthetic folic acid. It does not distinguish between biologically useful and potentially harmful forms. High serum folate may appear “normal” even if a toxic buildup is occurring.
Unmetabolised folic acid (UMFA):
This refers specifically to synthetic folic acid that has entered the bloodstream without being converted into its usable form (tetrahydrofolate). It reflects overload or enzyme bottleneck, not nutritional adequacy (Pfeiffer et al., 2015; Bailey and Ayling., 2009)
UMFA is not routinely measured in standard blood tests. It may accumulate silently, especially in people with low DHFR activity or MTHFR gene variants.
Think of serum folate as a cargo train carrying nutrients. Natural folate is processed and delivered at each stop. Synthetic folic acid (UMFA) however, stays on the train – unprocessed, piling up, and causing a traffic jam on the tracks.
6.2 Unmetabolised Folic Acid and functional Folate Deficiency
Numerous studies have shown that excessive intake of synthetic folic acid can disrupt the body’s natural folate metabolism. Under normal physiological conditions, folate is converted into its active form – 5-methyltetrahydrofolate (5-MTHF) – primarily in the liver. However, when synthetic folic acid is consumed in excess, particularly through fortified foods and supplements, this metabolic pathway becomes saturated. As a result, biologically inactive forms such as unmetabolised folic acid (UMFA) and dihydrofolate (DHF) may accumulate in circulation, leading to a condition termed functional folate deficiency (Cochrane et al., 2023; Smith et al., 2020). Serum blood tests may appear normal, but cells lack the activated folate they need (especially 5-MTHF). The buildup of unmetabolised folic acid (UMFA) in the bloodstream and has been shown to block folate metabolism and prevents DNA repair, methylation, and other critical functions (Troen et al., 2006; Smith et al., 2020; Cochrane et al., 2023; Puga et al., 2021).
Some biological consequences of UMFA and DHF accumulation include:
- Methylation disruption: Methylation is a key epigenetic mechanism that regulates gene expression. Disrupted folate metabolism can impair the methylation of DNA, RNA, proteins, and neurotransmitters, altering gene expression and cellular identity (Smith et al., 2020).
- Immune dysfunction: Elevated UMFA levels have been associated with suppression of natural killer (NK) cell activity, weakening the body’s innate immune response and increasing vulnerability to pathogens and chronic disease (Troen et al., 2006).
- DNA synthesis and repair impairment: UMFA and DHF interfere with the activity of enzymes responsible for DNA synthesis and repair, contributing to genomic instability and elevating the risk of mutations, cellular dysfunction, and carcinogenesis (Pfeiffer et al., 2015).
Critically, dihydrofolate reductase (DHFR) – the enzyme responsible for converting folic acid into tetrahydrofolate (THF) – has been shown to exhibit low and highly variable activity in human liver tissue. Excessive folic acid intake can overwhelm this enzyme, resulting in systemic accumulation of UMFA that cannot be metabolically converted into bioavailable folate forms (Bailey and Ayling, 2009).
This report will examine at length the many possible health implications of unmetabolised folic acid throughout; particularly for those with genetic vulnerabilities.
6.3 The CDC Perspective
The CDC continues to promote universal folic acid fortification as an unequivocal public health success, stating that “folic acid is the best tool to prevent neural tube defects” and that “all women of reproductive age should consume 400 mcg of folic acid daily” (CDC, 2023). However, these claims rest on outdated assumptions, omit significant risks, and disregard long standing evidence on genetic variation, metabolic impairment, and the long-term consequences of synthetic folic acid exposure.
Firstly, the assertion that folic acid is the optimal intervention ignores the growing body of evidence that natural methylated folates (e.g. L-5-methyltetrahydrofolate) may offer superior bioavailability, especially for individuals with impaired folate metabolism due to MTHFR or MTRR polymorphisms (Scaglione and Panzavolta, 2014). Many studies have proven that synthetic folic acid requires enzymatic activation that may be significantly compromised in populations with reduced MTHFR activity – raising questions about both efficacy and safety in these individuals (Bailey and Ayling, 2009).
Secondly, the CDC’s “one-size-fits-all” recommendation fails to account for interindividual variation in folate processing, sex-specific differences, or cumulative exposure via fortified food and supplements. Furthermore, despite folic acid being widely consumed in the United States and Canada, the CDC does not acknowledge the widespread presence of unmetabolised folic acid (UMFA) in the bloodstream. UMFA has been detected in over 95% of adults and is associated with potential adverse effects, including immune dysregulation, epigenetic alterations, and increased cancer risk in certain populations (Troen et al., 2006; Bailey et al., 2010).
The CDC’s folic acid resources also fail to include any substantive discussion of long-term outcomes, methylation pathways, or the potential interaction between synthetic folic acid and other micronutrients such as vitamin B12. Despite growing genetic evidence indicating that not all individuals metabolise synthetic folic acid effectively, public health guidelines have remained largely unchanged. This ongoing inaction raises serious concerns about systemic negligence – particularly in its disproportionate impact on genetically vulnerable populations, who are often left unprotected by one-size-fits-all policies.
CDC’s Unsubstantiated Claims on MTHFR and Folic Acid Fortification
Despite issuing categorical assurances that individuals with MTHFR gene variants “can process all types of folates, including folic acid,” the CDC fails to substantiate these claims with any robust scientific evidence (CDC, 2023). The CDC’s public guidance regarding MTHFR gene variants and folic acid supplementation references a narrow selection of sources, notably a general mechanistic review by Crider et al. (2012) and the OMIM (Online Mendelian Inheritance in Man) database. However, these sources do not provide direct empirical evidence supporting the safety of synthetic folic acid in individuals with impaired folate metabolism – particularly those homozygous for the C677T MTHFR variant, whose ability to convert folic acid to biologically active forms are significantly reduced (Crider et al., 2012; OMIM, 2024).
The Crider et al. review focuses primarily on the role of folate in DNA methylation and epigenetic regulation, with extensive discussion of biochemical pathways and theoretical mechanisms. While it affirms folate’s importance in early development, it does not distinguish between natural folate, synthetic folic acid, or L-methylfolate, nor does it assess population-level responses in MTHFR-compromised individuals (Crider et al., 2012). Moreover, it offers no clinical outcome data or longitudinal safety findings for this subgroup.
The OMIM entry on MTHFR is similarly descriptive – it outlines known polymorphisms and their biological significance, but does not reference interventional trials, toxicological data, or population-based outcomes related to folic acid exposure (OMIM, 2024). As such, the CDC’s reliance on these sources reflects a broader regulatory tendency to generalize safety claims without stratified evidence, thereby excluding genetically vulnerable populations from consideration.
This failure to provide adequate evidence constitutes a breach of scientific transparency and undermines the legitimacy of public health recommendations affecting genetically vulnerable groups. In contrast to this omission, independent research has consistently demonstrated that MTHFR variants can impair folic acid metabolism, potentially resulting in functional folate deficiency, neurodevelopmental impact, and increased risk of adverse outcomes when synthetic folic acid is consumed in excess or without adequate conversion (Lucock et al., 2014; Smith et al., 2008).
6.4 The NHS Position on Folic Acid and Genetic Vulnerability
The UK’s National Health Service (NHS) continues to recommend folic acid supplementation as a universal preventive strategy for neural tube defects (NTDs), advising all women who could become pregnant to take 400 micrograms daily until the twelfth week of pregnancy (NHS, 2024). High-risk individuals, such as those with diabetes or a family history of NTDs, are prescribed 5 mg of folic acid per day. However, this policy – echoed in the UK’s 2021 decision to begin mandatory fortification of non-whole meal wheat flour – lacks genomic sensitivity and fails to reflect emerging evidence regarding differential folate metabolism (UK Government, 2021).
Most notably, the NHS provides no public guidance regarding MTHFR gene polymorphisms, despite these being well-established genetic determinants of folate metabolism. Up to 14% of the UK population may carry homozygous (two copies) MTHFR C677T variants (Hazra et al., 2009), which significantly impair enzymatic conversion of synthetic folic acid into its bioactive form. The absence of any reference to these metabolic impairments constitutes a critical oversight, particularly in the context of population-wide fortification and high-dose prescription protocols.
In addition, the NHS does not engage with the extensive literature on unmetabolised folic acid (UMFA) accumulation. Studies in both the UK and the US have shown that individuals regularly exposed to fortified foods or supplements frequently exhibit detectable UMFA in their circulation, which has been associated with immune dysfunction, altered methylation patterns, and potential neurological harm (Troen et al., 2006; Bailey et al., 2010). The omission of this known biomarker of synthetic folate overload – particularly in those with MTHFR or MTRR impairments – reflects a failure to provide complete and balanced risk information.
Furthermore, no reference is made to L-5-methyltetrahydrofolate (5-MTHF) as a safer, biologically active alternative for individuals with impaired folate metabolism. This contradicts current best practice in clinical nutrition, where 5-MTHF is increasingly used to bypass metabolic bottlenecks in MTHFR-compromised patients (Scaglione and Panzavolta, 2014).
In conclusion, the NHS’s folic acid recommendations reflect a generalized, non-personalized model of public health that ignores genetic diversity, biochemical individuality, and long-term toxicological risks. Such omissions may violate informed consent principles and hinder the development of safer, precision-based nutritional policies.
6.5 Informed Consent and Genomic Oversight in Public Health Policy
Current NHS and CDC guidelines continue to recommend synthetic folic acid supplementation to all individuals of childbearing age, with no specific genomic stratification or reference to alternative folate forms such as L-5-methyltetrahydrofolate (5-MTHF), despite increasing evidence that individuals with MTHFR polymorphisms metabolize synthetic folic acid inefficiently. This omission not only lacks clinical nuance – it may constitute a breach of informed consent principles foundational to biomedical ethics.
According to Beauchamp and Childress’ framework (2013) and internationally recognized bioethical standards, valid informed consent must include:
- Disclosure of all relevant risks, benefits, and alternatives
- Comprehension of the information provided
- Voluntariness of the decision-making process
- Competence of the patient; and
- Explicit consent (Beauchamp and Childress, 2013; AMA Code of Medical Ethics, 2022).
Public health bodies are not exempt from this duty – particularly when interventions are mandated at a population level (e.g., through food fortification). Failure to disclose differential metabolic processing and potential adverse effects in genetically vulnerable subgroups undermines the autonomy of affected individuals. Furthermore, the omission of 5-MTHF as a clinically validated alternative (Gilbody et al., 2007; Scaglione & Panzavolta, 2014) may hinder the development of precision-based nutritional policy and constitutes a violation of the disclosure requirement embedded in informed consent.
The continued emphasis on mandatory fortification, despite the availability of safer, more bioavailable folate alternatives, raises questions about the influence of industry interests in shaping public health policy. The lack of open discourse around these alternatives suggests that convenience and corporate alignment may at times outweigh transparent, evidence-based decision-making.
6.6 Informed Consent, Bioethics, and the Systemic Omission of Genetic Risk
Unlike the United States, where the American Medical Association publishes a unified Code of Medical Ethics, the United Kingdom relies on a combination of legal precedent and professional standards to guide medical ethics. The General Medical Council’s (GMC) Good Medical Practice serves as the central ethical framework for UK-registered clinicians, outlining responsibilities related to informed consent, risk disclosure, and patient autonomy (GMC, 2024). While this guidance is influential and enforceable through professional disciplinary measures, it is not statutory law. However, the 2015 UK Supreme Court ruling in Montgomery v Lanarkshire Health Board marked a legal turning point, establishing that patients must be informed of any material risks of a proposed treatment and of reasonable alternatives (Montgomery v Lanarkshire Health board, 2015). This case reframed informed consent as a patient-centred right, rather than a clinician-determined obligation. In the context of population-level interventions such as folic acid fortification, failure to disclose genetic vulnerabilities (e.g. impaired folic acid metabolism in individuals with MTHFR polymorphisms) and safer alternatives (such as L-5-methyltetrahydrofolate) raises concerns about compliance with both ethical standards and the legal expectations defined by Montgomery. Despite these developments, UK public health policies continue to adopt a one-size-fits-all model, suggesting a gap between bioethical principles, legal precedent, and their application in preventive medicine.
Despite this precedent, the NHS continues to recommend universal folic acid supplementation and to support the UK government’s 2021 mandatory flour fortification policy without any acknowledgement of MTHFR gene polymorphisms or the availability of L-5-methyltetrahydrofolate (5-MTHF) as a viable, safer alternative. The NHS does not currently offer MTHFR testing or recognise the variant in clinical practice outside of a limited thrombophilia context, and no public patient guidance mentions differential folate metabolism or unmetabolised folic acid accumulation. While isolated research projects such as the CAREFOL-HT study have examined 5-MTHF in conditions like preeclampsia, these investigations remain siloed from public policy and do not constitute actionable patient information (HRA, 2021).
This omission represents more than a clinical oversight – it constitutes a systemic exclusion of a genetically identifiable subgroup from the protections of informed consent. Through failing to disclose both the potential for harm and the availability of an alternative, institutions such as the NHS may be in violation of the disclosure requirement at the heart of the Montgomery ruling. When applied at the population level, such omissions take on greater ethical gravity. They reinforce a one-size-fits-all model that not only disregards interindividual genetic variability, but may disproportionately expose vulnerable populations to risk. This gap between bioethical standards, legal expectations, and policy implementation raises serious concerns about regulatory negligence and epistemic injustice within state-sponsored nutrition programmes.
6.7 Eugenic Echoes and Epigenetic Risk in Genetically Stratified Populations
The failure to disclose genotype-specific risks within public health nutrition frameworks raises not only legal and ethical concerns but also ideological questions regarding the values embedded in policy design. When disregarding the metabolic vulnerability of individuals with MTHFR polymorphisms and omitting reference to established alternatives such as L-5-methyltetrahydrofolate (5-MTHF), UK health policy risks enacting a form of passive eugenics – not through active selection, but through selective negligence. The implicit assumption that a singular intervention (e.g. synthetic folic acid) is universally safe, despite growing evidence of harm in specific subgroups, reinforces a technocratic model in which genetically “noncompliant” bodies are rendered invisible, and their suffering unrecorded.
This systemic omission may contribute to epigenetic harm in populations already experiencing functional methylation impairments. As folic acid must be reduced via the MTHFR pathway before it becomes biologically usable, individuals with reduced enzyme activity may accumulate unmetabolised folic acid (UMFA), a compound associated with immune dysregulation, altered neurodevelopment, and carcinogenic potential (Troen et al., 2006; Reynolds, 2017). The epigenetic load imposed on these populations is not accidental – it is a predictable consequence of policy decisions made in the absence of genomic stratification. When the state enforces population-wide exposure to a synthetic nutrient without accounting for genetically variable processing, it perpetuates an outdated model of one-directional, top-down “health optimisation” that historically mirrored eugenic logic.
Modern public health must reckon with the genomic turn: the growing imperative to individualise intervention based on risk, resilience, and biochemistry. In ignoring this shift, institutions risk compounding disparities and eroding trust – particularly among populations historically excluded from medical authority. This is not merely a scientific failure; it is a failure of vision, ethics, and justice.
These omissions, when viewed through the lens of genetic exclusion and systemic harm, begin to reveal deeper ideological patterns. A broader analysis of this phenomenon -and its historical continuity with eugenic logic and soft bioengineering frameworks – is explored in Section 15: Eugenics, Bioengineering or Genocide?
7: Pregnancy, Infant Feeding and Related Risks (Also see Section 14.3 on Study by McNulty et al. (2013) and Section 9.2 on FRAA’s).

7.1 UMFA, Methylation Disruption, and Thrombotic Risk
Disrupted folate metabolism – particularly in the context of synthetic folic acid overexposure – has been increasingly implicated in vascular pathology. One key mechanism involves impaired methylation and the consequent elevation of plasma homocysteine, a known risk factor for venous thromboembolism (VTE). In individuals with polymorphisms such as MTHFR C677T or MTRR A66G, the enzymatic conversion of folic acid to its bioactive form (5-MTHF) is significantly impaired, leading to functional folate deficiency despite sufficient serum levels (Bailey & Ayling, 2009). Elevated homocysteine concentrations have been consistently associated with increased risk of deep vein thrombosis, pulmonary embolism, and cerebrovascular accidents (den Heijer et al., 1996; Lentz, 2005). Moreover, excess UMFA may disrupt endothelial integrity and promote oxidative stress, impairing nitric oxide bioavailability and exacerbating vascular inflammation (Troen et al., 2006). These combined mechanisms contribute to a prothrombotic state, which may be further exacerbated by external factors such as hormonal contraceptive use, pregnancy-related immobility, and elevated homocysteine levels, particularly in individuals with impaired methylation capacity (ASRM, 2016; CDC, 2023; Selhub, 1999). In such cases, excessive exposure to synthetic folic acid – when unmetabolised due to MTHFR polymorphisms – may fail to reduce homocysteine effectively, thereby intensifying thrombotic risk rather than mitigating it.
7.2 Endothelial Dysfunction and Folic Acid-Linked Vascular Compromise in Pregnancy
In the context of pregnancy, optimal vascular health is essential for placental perfusion, nutrient exchange, and foetal development. However, excessive synthetic folic acid intake may impair these processes by contributing to endothelial dysfunction and microvascular injury. Unmetabolized folic acid (UMFA) has been shown to accumulate in the plasma when intake exceeds the liver’s enzymatic capacity, particularly in individuals with sluggish dihydrofolate reductase (DHFR) activity (Bailey & Ayling, 2009). This accumulation may interfere with folate receptor binding, transport, and downstream methylation pathways, leading to epigenetic instability and immune dysregulation (Obeid et al., 2015). Furthermore, elevated homocysteine – a common result of impaired folate-dependent methylation – has been directly associated with placental vascular lesions, pre-eclampsia, recurrent miscarriage, and intrauterine growth restriction (IUGR) (Zhang et al., 2004; Ueland et al., 2000).
Several studies have reported associations between elevated levels of unmetabolised folic acid (UMFA) and an increased risk of miscarriage. Obeid et al. (2015) identified high UMFA concentrations as a potential contributor to adverse pregnancy outcomes, including early pregnancy loss. Similarly, Yin et al. (2012) observed increased miscarriage rates among women receiving excessive folic acid supplementation in the first trimester. Although causality remains contested, emerging research highlights broader concerns surrounding folate imbalance and genomic instability during gestation. High folic acid intake has been linked to chromosomal fragility in peripheral lymphocytes (Colapinto et al., 2022), while an imbalanced folate-to-B12 ratio – a common outcome of excess folic acid exposure – has been associated with miscarriage, preeclampsia, and foetal growth restriction (Naseh et al., 2024). Furthermore, high-dose folic acid supplementation (≥800 μg/day) has been implicated in increased rates of gestational hypertension, a condition known to elevate miscarriage and placental abruption risks (Zhao et al., 2019).
Further studies also suggest that excess synthetic folic acid may contribute to adverse pregnancy outcomes, particularly in genetically susceptible populations. Women with homozygous MTHFR C677T variants, who already experience impaired conversion of folic acid to its bioactive form, may paradoxically be at greater risk of functional folate deficiency, even in the presence of high serum folate. Several studies have reported associations between elevated unmetabolised folic acid (UMFA) levels and increased risk of miscarriage, placental abnormalities, and gene expression dysregulation in placental and embryonic tissues (Obeid et al., 2015; Yin et al., 2012). Additionally, folic acid supplementation without consideration of gene-environment interactions may obscure the nuanced role of folate cycle balance, potentially shifting the uterine environment toward epigenetic instability and immune dysregulation (Bailey & Ayling, 2009; Lucock et al., 2013).
These findings suggest that, in cases of impaired metabolism, synthetic folic acid may paradoxically compromise the very pregnancy outcomes it was intended to support. A precision-medicine approach, incorporating genetic screening and methylation support, is urgently needed to address this overlooked iatrogenic risk.
7.3 The Manufactured Silence: Suppression of Research into Folic Acid and Pregnancy Loss
Despite mounting mechanistic, observational, and biochemical evidence suggesting that excess unmetabolised folic acid (UMFA) may contribute to adverse pregnancy outcomes – including miscarriage – there remains a conspicuous absence of large-scale, causally oriented research exploring this link directly. While studies such as Obeid et al. (2015) and Yin et al. (2012) have reported associations between high folic acid intake and early pregnancy loss, public health authorities continue to frame folic acid as universally safe. This is not due to a wealth of exonerating evidence – but rather, pharmaceutical led engineering, data manipulation and a lack of relevant investigation – in order to maintain control on the ‘safe’ narrative.
The failure to pursue definitive studies in this area is not a scientific oversight – it is a deliberate preservation of plausible deniability. If causality were established – if even one well-powered, peer-reviewed study confirmed that excess folic acid contributes to miscarriage – it would trigger a seismic reckoning across global nutrition policy, medical ethics, and pharmaceutical regulation. Such a revelation would not only undermine decades of public health messaging, but also expose regulators and institutions to legal liability for promoting or mandating a compound now shown to have genotype-specific toxicity.
Instead, what has emerged is a textbook example of manufactured uncertainty – a suppression model well-documented in the histories of leaded petrol, tobacco, and asbestos (Michaels, 2008). Through avoiding causal research, health authorities and funders preserve institutional deniability while continuing to expose the public to risk. This is not scientific caution – it is institutional self-protection masquerading as evidence-based policy.
That governments continue to promote folic acid supplementation and fortification as a blanket policy – without genetic stratification, informed consent, or transparent risk disclosure – despite the possibility of contributing to pregnancy loss, reflects a systemic betrayal of ethical responsibility. When the tools to uncover truth are readily available but remain unused, silence becomes complicity, and plausible deniability becomes policy.
7.4 Empirical Evidence: Breast Milk, Population Studies, and Enzyme Disruption
A 2023 study by Cochrane et al. (Scientific Reports) reported that mothers supplementing with synthetic folic acid had UMFA levels comprising 28% of total milk folate, compared to just 2% in those using natural folate forms such as 5-MTHF. This significant disparity raises important questions about infant exposure to UMFA during critical stages of development, particularly in relation to methylation processes and immune system maturation.
In a 2015 study published in The Journal of Nutrition, Pfeiffer et al. found that over 95% of individuals in the U.S. had detectable levels of UMFA in their bloodstream, suggesting widespread exposure resulting from fortification and supplementation practices. Notably, elevated concentrations were most prevalent among non-fasting individuals, older adults, supplement users, and non-Hispanic black populations. These findings were further associated with suppressed natural killer (NK) cell activity, indicating potential implications for immune function. This emerging evidence highlights the critical role of maternal immune integrity in shaping the infant immune system, particularly via breastfeeding. NK cells serve as a frontline defence against viral infections and tumours, and their suppression may have systemic immunological consequences.
A 2022 study confirmed that prolonged intake of high-dose folic acid (5 mg/day for 90 days) significantly increased UMFA while reducing NK cell cytotoxicity in healthy adults (Huang et al., 2022). Similarly, preclinical models have shown that excess folic acid impairs NK cell-mediated immune surveillance (Hibbs et al., 2016). These findings underscore a broader concern: excess folic acid may not be immunologically inert, especially when unmetabolised forms accumulate.
Breast milk is not merely nutritional; it is an immunological extension of the mother, providing leukocytes, secretory antibodies, cytokines, and bioactive compounds that influence the development of the infant’s immune system (Khan et al., 2022). If maternal immune cell function is altered – such as through folic acid–induced NK suppression – it may impair the immunity protecting quality of breast milk. Studies have shown that breastfeeding enhances thymus size and immune competency in infants, compared to formula feeding (Prentice et al., 2002). Disruption to this immunological transfer, even indirectly via maternal micronutrient metabolism, deserves urgent attention.
Although direct studies linking UMFA to impaired immune transfer via breast milk are lacking, the mechanistic plausibility is strong and grounded in existing human and animal studies. In a vulnerable newborn, any reduction in maternal immune protection may have disproportionate effects, particularly in the critical first six months of life.
Research published by Puga et al. (2021) published in Nutrients corroborates the concern that high levels of synthetic folic acid may inhibit critical enzymes such as dihydrofolate reductase (DHFR) and methylenetetrahydrofolate reductase (MTHFR), thereby limiting conversion to bioactive folate forms. This enzymatic inhibition has been linked to oxidative stress, DNA strand breaks, and diminished repair capacity, particularly in individuals with MTHFR gene variants – raising important questions about the long-term safety of high-dose folic acid intake.
In summary, the MTHFR gene encodes an enzyme vital to converting synthetic folic acid into its bioactive form, 5-methyltetrahydrofolate (5-MTHF). This conversion is essential for key methylation processes, including DNA repair, gene regulation, and neurotransmitter synthesis. Individuals with the homozygous C677T mutation (TT genotype) demonstrate up to a 70% reduction in enzymatic activity compared to those with the typical (CC) genotype – a difference with considerable implications for folate metabolism and functional sufficiency (Obeid & Herrmann, 2019).
7.5 Synthetic Folic Acid in Infant Formula: Risks Associated with UMFA Accumulation and MTHFR Gene Mutations
The widespread inclusion of synthetic folic acid in infant formula is an underexplored health risk, particularly for infants with genetic polymorphisms that impair folate metabolism. Folic acid, a synthetic oxidized form of vitamin B9, must be reduced by dihydrofolate reductase (DHFR) and further converted by methylenetetrahydrofolate reductase (MTHFR) to its biologically active form, 5-methyltetrahydrofolate (5-MTHF) (Bailey & Ayling, 2009; Crider et al., 2011). However, both neonates and individuals with MTHFR gene mutations – such as the C677T and A1298C variants – show a reduced capacity to carry out these conversions (Weisberg et al., 1998; McNulty et al., 2000).
Formula-fed infants receive folic acid as their sole folate source, unlike breastfed infants, who obtain natural folate through human milk (Green et al., 2005). This results in significantly higher plasma folate and unmetabolised folic acid (UMFA) concentrations in formula-fed infants, a pattern that is concerning given the immature metabolic systems of neonates (O’Connor et al., 2013). DHFR activity in human infants is markedly low and variable, leading to inefficient conversion of folic acid and the accumulation of UMFA in circulation (Bailey & Ayling, 2009; McNulty et al., 2000; Pfeiffer et al., 2015).
Formula-fed infants receive folic acid as their sole folate source, unlike breastfed infants who receive natural reduced folates through human milk; this distinction is critical. A study by Green et al. (2005) reported that plasma folate and UMFA levels were significantly higher in formula-fed infants, raising immediate concern given the immature metabolic capacity of neonates. Further, O’Connor et al. (2013) and Bailey et al. (2009) found that dihydrofolate reductase (DHFR) activity in human infants is both markedly low and variable, resulting in poor conversion of folic acid and leading to systemic accumulation of unmetabolised folic acid (UMFA) in circulation.
This early and sustained exposure to high levels of synthetic folic acid – during a critical window of immune and cellular development – raises red flags, particularly for infants with MTHFR gene polymorphisms, who have reduced capacity to generate biologically active folate. Animal and adult studies have linked elevated UMFA to genomic instability, disrupted methylation, and immune suppression, including diminished NK cell cytotoxicity, which is vital for early-life tumour and infection defence (Hibbs et al., 2016; Obeid et al., 2015; Pfeiffer et al., 2015).

This is not a theoretical risk. It is an uncontrolled, early-life exposure to a synthetic compound that bypasses normal enzymatic filters and builds up in developing systems. In this context, current infant formula practices – while intended as nutritional support – may constitute an unexamined toxicological burden.
Given that childhood cancer incidence is rising, this is no longer a hypothetical concern. In the United Kingdom, childhood cancer rates have increased by 8% since the early 2000s, with the trend most pronounced among boys (Cancer Research UK, 2024). In the United States, data from the Surveillance, Epidemiology, and End Results (SEER) program show a consistent annual increase of approximately 0.5% in paediatric cancers from 2003 to 2019, with sharper rises between 2003 and 2016 (Thomas et al., 2023). These increases are occurring too rapidly to be attributed to genetics alone, suggesting environmental, nutritional, or pharmaceutical contributors that are yet to be adequately examined.
UMFA accumulation has been implicated in a range of adverse effects, including disruption of immune function, reduced natural killer cell activity, and interference with methylation-dependent gene expression (Troen et al., 2006; Kalmbach et al., 2008). Natural killer (NK) cells serve as a frontline defence against viral infections and malignant transformation, detecting and eliminating infected or abnormal cells without prior sensitisation (Vivier et al., 2021; Zhao et al., 2024). Their suppression may have systemic immunological consequences, particularly in neonates and immunologically naïve populations who rely heavily on maternal immune components for protection.
These effects may be further intensified in infants with homozygous MTHFR polymorphisms, which reduce enzyme function by up to 70%, compromising folate cycling and methylation (Scaglione & Panzavolta, 2014). Chronic exposure to synthetic folic acid during this critical developmental window may thus impose a disproportionate burden on infants with these polymorphisms, leading to epigenetic consequences.
7.6 Epigenetic risk in Pregnancy
Emerging animal and human studies suggest that early-life exposure to excess synthetic folic acid can result in epigenetic reprogramming, with potential long-term health effects. Sweeney et al. (2007) demonstrated that synthetic folic acid supplementation during gestation in rats altered DNA methylation patterns in offspring. Similarly, Waterland and Jirtle (2003) showed that folate availability during early development influenced transposable element methylation and gene expression across the lifespan. These findings imply that excessive folic acid exposure may imprint lasting changes on infant gene regulation, especially in methylation-sensitive regions.
A further concern is the presence of folate receptor alpha autoantibodies (FRAAs) in children with neurodevelopmental differences, including autistic children (See also Section 9.2 on FRAA’s). These autoantibodies block folate transport across the blood–brain barrier and are associated with cerebral folate deficiency syndromes, even when dietary intake is sufficient (Ramaekers et al., 2007). It is hypothesised that UMFA accumulation or overexposure to synthetic folic acid may trigger FRAA production or exacerbate neurological vulnerability in genetically susceptible infants.
Given that MTHFR variants affect a significant portion of the global population – up to 40–60% in some ethnic groups (Weisberg et al., 1998) – the current reliance on synthetic folic acid in infant formula may represent a widespread but unrecognised risk factor for immunological and neurodevelopmental outcomes. Reformulating infant formulas to include 5-MTHF or natural folate, rather than synthetic folic acid, could significantly reduce the burden of UMFA exposure and accommodate the biochemical individuality of infants.

8: Toxicity -The Threshold That Doesn’t Exist: How UMFA Remains Officially Undefined
Despite overwhelming evidence that unmetabolised folic acid (UMFA) is present in most individuals tested – often over 90% in fasted subjects (Pfeiffer et al, 2015) – there remains no official safety threshold or biomarker classification for UMFA in public health policy. This is not a scientific limitation; it is a structural strategy: by refusing to define what level of UMFA is considered toxic or actionable, regulators and industry-aligned scientists can acknowledge its presence while denying its relevance.
UMFA is a biochemical signal of system overload. It represents synthetic folic acid that has not been converted by the liver into usable forms of folate. In individuals with impaired methylation capacity – such as those with MTHFR polymorphisms – this unmetabolised folic acid can build up in the bloodstream, where it interferes with folate receptor binding, methylation cycles, immune function, and potentially tumour suppression (Troen et al, 2006; Cochrane et al, 2023).
Yet, despite these well-documented risks, UMFA is not recognized in routine clinical testing. There is no recommended upper limit and no established cut-off value. – no agreed-upon intervention threshold. This silence is strategic; by failing to define UMFA as a biomarker of concern, regulators can claim that there is ‘no evidence of harm’ – because no one is officially measuring it.
In toxicology, this omission would be considered gross negligence; in public health, it becomes policy insulation. The absence of a UMFA threshold is not an oversight – it is a firewall, and behind it, a generation of vulnerable individuals continue to accumulate synthetic folic acid their bodies cannot use or expel. Those with MTHFR mutations, impaired methylation, and systemic folate metabolism disorders – those most impacted by synthetic folic acid – have been mathematically removed from the dominant narrative. Their biology complicates the narrative model, so it is ignored. However, the presence of unmetabolised folic acid (UMFA) in over 90% of fasted test subjects proves that systemic exposure exists. The failure to define a toxicity threshold for UMFA is not scientific caution – it is policy protection.
Folate plays a critical role not only in DNA methylation but also in histone modification, which governs chromatin structure and accessibility of gene expression – especially in the developing brain (Crider et al, 2012). Histone methylation and acetylation are dynamic processes that affect synaptic plasticity, neurogenesis, and cognitive flexibility. Aberrant folate metabolism, particularly under conditions of synthetic folic acid overload, can disrupt the availability of methyl donors such as S-adenosylmethionine (SAM), leading to histone hypomethylation and impaired neural regulation (Zeisel, 2009; Blusztajn & Mellott, 2012). In experimental models, folate imbalance has been shown to alter expression of genes involved in neurotransmitter systems, learning, and memory consolidation, while prenatal folate perturbations have led to long-lasting behavioural effects (Konycheva et al., 2011; Craciunescu et al., 2004). These findings indicate that excessive or improperly metabolized folic acid may distort the neuroepigenetic landscape, particularly in vulnerable periods of development, potentially contributing to neurological ‘disorders’, ADHD, and other neuropsychiatric conditions through histone-mediated mechanisms.
One of the most strategically deceptive elements in the discourse on maternal nutrition and neurodevelopmental outcomes has been the persistent mislabeling of synthetic folic acid exposure as “folate.” This semantic manipulation has enabled public health authorities and medical institutions to obscure the damaging effects of unmetabolized folic acid (UMFA), while maintaining the appearance of nutritional advocacy. The implications of this deliberate conflation are profound, as it has facilitated mass exposure to a synthetic compound under the guise of an essential nutrient, and has hidden critical data from vulnerable populations.
A 2016 press release from Johns Hopkins Bloomberg School of Public Health illustrates this obfuscation with disturbing clarity. The article, titled “Too Much Folate in Pregnant Women Increases Risk for Autism, Study Suggests,” received broad media coverage and has been cited as a turning point in the discussion around prenatal supplementation. However, the article fails to clarify that the biomarker used in the study was not dietary folate or total folate levels, but specifically UMFA—a synthetic byproduct that accumulates in maternal and fetal circulation following high intake of folic acid.
The source study itself, later published in full as Raghavan et al. (2020), makes this distinction clear: “The association with ASD was found only for cord blood UMFA concentrations, not for 5-methyltetrahydrofolate (5-MTHF), nor for total folate.” (Raghavan et al., 2020). Despite this, the public-facing summary replaces “unmetabolized folic acid” with the generic term “folate” throughout, effectively dissolving the possibility of public accountability.
This linguistic sleight of hand has a twofold effect. First, it absolves public health authorities of the need to reassess blanket folic acid fortification policies, which were instituted in the late 1990s despite the lack of safety data for high-dose, long-term exposure. Second, it perpetuates confusion among healthcare professionals and the public, many of whom remain unaware of the biochemical difference between folic acid and naturally occurring folate derivatives such as 5-MTHF.
The Johns Hopkins article, and others like it, serve as a form of controlled disclosure: a limited acknowledgment of harm, carefully buffered by misdirection. By revealing just enough data to retain scientific credibility, but framing it with deliberately vague language, the narrative protects institutional credibility while compromising public trust. It also diverts scrutiny away from vulnerable populations, such as individuals with MTHFR gene variants, who are less able to convert folic acid into usable forms and are thus disproportionately affected by UMFA toxicity.
It is also essential to critically challenge the framing of outcomes like “autism” in these contexts. What these studies are observing and labeling as “autism risk” is more accurately understood as neurological injury -environmentally induced, preventable damage that disproportionately affects those with underlying neurodivergent biology. Many of the individuals harmed may already be autistic, or genetically predisposed to a sensitive neurotype, and the true effect is not the “creation” of autism but the exacerbation or distortion of neurodevelopment through toxic insult. The language used in such publications falsely implies that autism itself is a defect caused by exposure, rather than acknowledging that what is being documented is injury to autistic people via systemic environmental poisoning.
The consequences of this manipulation are not merely theoretical. The study in question found that mothers with high UMFA levels at delivery had significantly elevated odds of having a child diagnosed with autism spectrum disorder (ASD). When both UMFA and B12 were elevated, the odds ratio increased to 17.6 -a risk magnitude that should have triggered immediate international review of folic acid policy. Instead, it was hidden in plain sight and distorted through diagnostic oversimplification.
Additional evidence from randomized trials further reinforces these concerns. In a landmark double-blind study conducted in Norway -a country without folic acid fortification -Ebbing et al. (2009) found that daily supplementation with 0.8 mg folic acid and 0.4 mg vitamin B12 was associated with a 21% increased risk of cancer incidence, 38% increased cancer mortality, and 18% increase in all-cause mortality. These results emerged from a cohort of over 6,800 patients with ischemic heart disease and represent one of the most robust human trials demonstrating the long-term dangers of synthetic folic acid. The findings cannot be dismissed as regional anomalies; rather, they call into question the safety of high-dose folic acid in any context.
It is essential to clarify that the term “neurological disorders” as used in this context may obscure a deeper, more insidious truth. Many of the ‘severe’ challenges associated with neurodivergent populations -such as autistic people, those with ADHD, or sensory integration differences -may not be inherent “disorders,” but rather manifestations of neurological harm in genetically or epigenetically vulnerable individuals. In particular, individuals with MTHFR or related polymorphisms may be more susceptible to neuroepigenetic disruption when exposed to high levels of synthetic folic acid or other metabolic stressors during critical developmental windows. As such, the line between “disorder” and iatrogenic injury is not merely academic – it is political, medical, and ethical. Recognising this distinction is crucial, as it shifts the lens from pathologising neurodivergent identities to exposing the structural and pharmaceutical conditions that may distort, suppress, or injure them.
Regulatory bodies such as the EFSA and FDA have established tolerable upper intake levels (ULs) for folic acid, primarily to prevent vitamin B12 deficiency masking (EFSA, 2006; IOM, 1998). Yet they have never established a serum UMFA toxicity threshold. Despite mounting concerns about UMFA’s links to impaired immune function, altered methylation, and potential tumour proliferation, agencies cite ‘insufficient evidence’ as the reason for inaction. This inaction ensures the cycle continues unmeasured, unregulated, and unchecked.
A toxicological framework that fails to define what toxicity looks like is not protection – it is plausible deniability. The absence of UMFA thresholds keeps public health policies insulated from liability, while millions are subjected to a biochemical exposure they cannot detoxify. This is not an oversight; it is omission as strategy.
8.1 Toxic Break Down
A 2021 study in ‘Free Radical Biology and Medicine’ (Goossens et al., 2021) synthetic folic acid does not break down efficiently in the body, which can lead to the formation of harmful byproducts such as 6-formylpterin (6-FPT). These byproducts have been linked to serious health risks, including:
- Oxidative stress and DNA damage, which can increase the risk of cancer development by destabilizing genetic material (Goossens et al., 2021).
- Immune dysfunction, as synthetic folic acid has been found to interfere with mucosal-associated invariant T (MAIT) cells, which play a crucial role in immune response (Thuru et al., 2022).
- Neurodegeneration, as unmetabolized folic acid (UMFA) can accumulate in the body, potentially contributing to cognitive decline and neurological damage (Bailey et al., 2010).
In contrast to natural folate, synthetic folic acid is highly unstable under environmental conditions such as heat, light, and prolonged storage. Both cooking and baking degrade its structure, reducing its bioavailability. Moreover, when exposed to these conditions, synthetic folic acid may break down into oxidative byproducts that further contribute to cellular stress and genomic damage (Goossens et al., 2021). This instability not only compromises its efficacy but may also introduce additional biological risks over time.

8.2 Alternatives: Folate Form Comparison
The table below compares three primary forms of folate – folic acid, folinic acid (Leucovorin), and methylfolate (5-MTHF)- with regard to their biochemical behaviour, safety profiles, and clinical applications. This comparison illustrates why folinic acid and methylfolate are considered safer and more biologically appropriate options, particularly for individuals with known genetic susceptibilities.
| Feature | Folic Acid | Folinic Acid | Methylfolate (5-MTHF) |
| Chemical Name | Pteroylmonoglutamic acid | 5-formyl tetrahydrofolate | 5-methyltetrahydrofolate |
| Natural or Synthetic | Synthetic (Bailey & Ayling, 2009) | Natural derivative (Moretti et al., 2005) | Bioactive natural form (Crider et al., 2012) |
| Requires DHFR Conversion | Yes (slow and variable) (Obeid et al., 2015) | No | No |
| Produces UMFA | Yes (Crider et al., 2012) | No | No (Blusztajn & Mellott, 2012) |
| Supports Methylation | Indirectly, if converted (Obeid et al., 2015) | Yes (Moretti et al., 2005) | Yes (directly) (Roffman et al., 2011) |
| Safe for MTHFR Mutations | No – may be harmful | Yes – generally well tolerated | Yes – optimal for MTHFR |
| Crosses Blood-Brain Barrier | Poorly (Obeid, R. and Herrmann, W., 2012) | Moderate | Good (especially as calcium salt) (Crider et al., 2012) |
| Cost (approximate) | Very low | Moderate | Moderate to high |
| Common Use | Fortified foods, cheap supplements | Therapeutic use, neuroprotection | Targeted supplements, therapeutic methylation |
Despite mounting scientific evidence highlighting the risks associated with synthetic folic acid and the availability of safer, naturally occurring alternatives such as 5-MTHF, these options remain underutilised and largely absent from mainstream fortification strategies. Instead, synthetic folic acid continues to dominate public health policy -often prioritised due to cost-efficiency and commercial interests, rather than robust evidence-based safety considerations.
9: Not The Cause – The Signal: Reframing Autism in a Toxic World
Historically, miners would carry canaries into coal mines to serve as early detectors of toxic gases; their heightened sensitivity provided a warning system before danger became fatal. In a strikingly parallel manner, autistic individuals often exhibit increased sensitivity to environmental toxins, synthetic additives, and dietary components (Rossignol & Frye, 2014). This sensitivity is not a pathology – it is biological, genetic, and intrinsic. It positions autistic people not as anomalies, but as early warning sentinels -individuals whose physiology responds first and most clearly to the environmental pressures that may affect us all (Rossignol & Frye, 2014; Landrigan, 2010; Stamova et al., 2011).
A key factor in this context is the MTHFR gene, which affects folate metabolism. Variants such as the C677T mutation impair the body’s ability to process synthetic folic acid efficiently (Obeid & Herrmann, 2012). Studies have shown that this genetic mutation appears at significantly higher rates in the autistic population compared to neurotypical controls (Li et al., 2020). This altered metabolism contributes to elevated homocysteine levels and accumulation of unmetabolised folic acid (UMFA), both of which may have systemic effects.
To return to the metaphor – when a canary succumbs, miners do not blame the bird. They recognise the air is poisoned; and yet, when autistic individuals experience chronic illness, neurological dysregulation, or metabolic impairment in response to environmental factors -the response is too often diagnostic rather than environmental -pathologising the person as disordered rather than interrogating the toxic context.
9.1 Autism, MTHFR, UMFA, and the Invisible Epidemic
Recent data suggest that a significant anount of autistic individuals carry at least one MTHFR mutation, though peer reviewed data is sparse (Li et al., 2020). This places them at a significant metabolic disadvantage when exposed to synthetic folic acid, impairing their ability to convert it to its usable form. The consequences may include folate trapping, methylation dysfunction, and cascading impacts on neurotransmitter synthesis, gene expression, and detoxification capacity.
In broader populations, approximately 10–15% of individuals in North America and Europe are homozygous for the MTHFR C677T mutation, meaning they inherit the gene from both parents (Frosst et al., 1995; Botto & Yang, 2000). This results in significantly reduced enzyme activity, leading to increased vulnerability to UMFA accumulation and associated neurological and immunological disruptions (Bailey & Ayling, 2009).
Multiple studies across international populations have found an elevated prevalence of the homozygous C677T MTHFR mutation in autistic people. For instance, a study of the Chinese Han population reported the TT genotype in 16.1% of autistic children, compared to just 8.6% among matched controls (Pu et al., 2023). Similarly, a Turkish cohort identified the TT genotype in 3.1% of autistic children, while it was not detected in any participants from the control group (Pu et al., 2023). Though the frequency varies across ethnicities and environments, the pattern of increased MTHFR mutation prevalence among autistic individuals is consistent and merits closer investigation – particularly in the context of widespread synthetic folic acid exposure.
9.2 Folate Receptor Alpha Autoantibodies (FRAAs)
FRAAs are autoantibodies that bind to the folate receptor alpha (FRα) – a key receptor responsible for transporting folate across the blood–brain barrier. When these autoantibodies are present, they block or reduce folate transport into the cerebrospinal fluid (CSF), which can lead to a functional cerebral folate deficiency, even when blood folate levels are normal (Cochrane et al., 2023; Troen et al., 2016).
Folate receptor alpha autoantibodies (FRAAs) have been detected in approximately 70-75% of autistic children, particularly in autistic individuals with additional neurological symptoms (Ramaekers et al., 2007; Frye et al., 2013). These autoantibodies block the transport of 5-methyltetrahydrofolate (5-MTHF) across the blood–brain barrier, leading to a functional cerebral folate deficiency – even when serum folate levels appear normal. This disruption in folate delivery to the central nervous system has been linked to motor dysfunction, white matter abnormalities, and cognitive impairment. Despite this, FRAAs remain virtually absent from public health discourse. Their presence has been speculated as a potentially treatable subset of autism, yet current policy and fortification strategies ignore this biomarker entirely, subsequently denying affected children access to individualised, targeted intervention.
9.3 Folate Receptor Alpha Autoantibodies (FRAAs) as a Potential Marker of Neurological Injury in Autism
As stated, folate receptor alpha autoantibodies (FRAAs) have been detected in up to 70–75% of children diagnosed with autism spectrum disorder (ASD), compared to less than 10% in neurotypical controls (Frye et al., 2013; Ramaekers & Sequeira, 2021). FRAAs inhibit the binding of folate to its primary transporter into the central nervous system – the folate receptor alpha (FRα) – disrupting folate transport across the choroid plexus and resulting in cerebral folate deficiency (CFD), despite normal serum folate levels (Ramaekers et al., 2002; Vincent et al., 2011).
The presence of FRAAs is strongly associated with neurological regression, seizures, speech loss, and other neuroimmune symptoms in children with ASD, suggesting that their role is not incidental but mechanistically significant. FRAAs impair methylation, myelination, neurotransmitter synthesis, and immune signalling, contributing to neuroinflammation and altered brain development during critical periods (Frye et al., 2013; Rossignol & Frye, 2012).
Moreover, children with FRAAs often show evidence of mitochondrial dysfunction, glutathione depletion, and oxidative stress – patterns that align with toxicological injury and impaired redox balance. This suggests that FRAAs may be biochemical markers of environmental injury in genetically vulnerable individuals, particularly those with methylation-related gene polymorphisms such as MTHFR (Frye et al., 2016; Obeid et al., 2015).
While some researchers have framed this as a “treatable subtype” of autism, an alternative interpretation is this: these neurodivergent individuals may have sustained neurological injury through folate receptor autoimmunity triggered by environmental, pharmaceutical, or nutritional exposures. As such, FRAAs should not be viewed solely as a diagnostic subtype, but as a potential iatrogenic or epigenetic injury signal, in addition to their neurotype.
A growing body of research suggests that unmetabolised folic acid (UMFA) may exert neurotoxic effects, particularly in individuals with impaired folate metabolism. In populations carrying MTHFR and MTRR mutations, excess synthetic folic acid may contribute to disruptions in methylation, mitochondrial dysfunction, impaired detoxification, and increased oxidative stress – all mechanisms implicated in autism pathophysiology (Rossignol & Frye, 2014; James et al., 2004).
Several studies have observed that autistic individuals often present with elevated homocysteine levels, reduced glutathione activity, and poor folate transport across the blood-brain barrier (Quinlivan & Gregory, 2008; Frye et al., 2013). These factors suggest that the autistic brain may be particularly vulnerable to chronic UMFA accumulation and related biochemical stressors – especially during prenatal and early postnatal development when neural plasticity is at its peak.
Together, these findings raise compelling questions about the long-term neurodevelopmental impact of synthetic folic acid exposure – particularly in individuals with known genetic susceptibilities. They do not imply causation, but they challenge the assumption that synthetic folic acid is universally safe or biologically inert.
FRAAs in Infants and Children
- The brain relies heavily on folate during development, especially for myelination, DNA methylation, and neurotransmitter synthesis (Krishnaveni and Srinivasan, 2021).
- FRAAs can prevent 5-MTHF from reaching the brain, leading to neurological symptoms and developmental delays (Ramaekers et al., 2005).
- High levels of synthetic folic acid have been suggested to contribute to autoimmunity, possibly triggering FRAA production in susceptible individuals (Obeid and Herrmann, 2012; Quadros, 2015).
FRAAs and Autism
- Studies by Ramaekers et al. (2007, 2013) found elevated FRAAs in a significant proportion of autistic children (Ramaekers et al., 2007; Ramaekers and Rothenberg, 2013).
- Treating these children with high-dose Leucovorin (folinic acid) – which bypasses the blocked receptor -led to notable improvements in language, behavior, and cognition in some cases (Frye et al., 2013).
- It has been suggested that FRAAs may represent a ‘treatable subtype’ of autism that is being completely ignored in public health and nutritional policy discussions (Frye et al., 2018).
Synthetic folic acid accumulates as UMFA, especially in people with MTHFR mutations.
- There’s a speculative theory that chronic high folic acid intake may contribute to immune dysregulation and autoantibody production, possibly including FRAAs (Blaylock, 2008).
- This is especially concerning in infants fed synthetic folic acid–fortified formula, who may be epigenetically or immunologically primed to develop these issues (Sequeira et al., 2016).
9.4 MTHFR and the Neurodevelopmental Genetic Cluster
Many of the gene variants associated with the diagnostic term ‘autism spectrum disorder’ (ASD) – including MTHFR, COMT, SLC6A4, DRD4, and BDNF – are not exclusive to autism. Rather, they form part of a shared genetic cluster that is also implicated in neurodivergent conditions such as ADHD, schizophrenia, bipolar disorder, depression, and anxiety (Cross-Disorder Group of the Psychiatric Genomics Consortium, 2013). This overlap reflects a spectrum of neurological sensitivities rather than isolated “disease genes.”
The MTHFR gene, in particular, plays a pivotal role in methylation, folate metabolism, and neurotransmitter synthesis. Mutations in MTHFR, such as the C677T and A1298C polymorphisms, are associated with altered neurodevelopmental trajectories, especially under conditions of environmental or nutritional stress – like exposure to environmental toxins such as synthetic folic acid or unmetabolised folic acid (UMFA) buildup.
What this means is that MTHFR mutations don’t “cause autism”- they create a biochemical vulnerability. When combined with oxidative stressors, immune activation, or toxin exposure (e.g. during pregnancy or early infancy), these vulnerabilities may express as neurodevelopmental injury – interpreted as autism, ADHD, or other psychiatric conditions, depending on timing, severity, and context.
So, when researchers label certain genes as “autism risk genes,” they are often misrepresenting a complex web of shared neurodevelopmental pathways. These genes are not pathological – they are part of neurodivergent identity and may only become pathological in the presence of environmental triggers or systemic failure.
Processed foods, fortified grains, pharmaceuticals, pesticides, water fluoridation, plastics, EMFs – this chemical soup is tolerated by some, but it floods and destabilizes others. Neurodivergent people react with their bodies and brains, in a way that the rest of the population may take decades to feel.

Neurodivergent people are not defective, they are biochemically different – and the modern toxic environment is not designed for them. If autistic people – or those with homozygous MTHFR gene mutations are the early warning system, then every time their signals are suppressed, their expressions medicated, or their sensitivities gaslit, society silences the truth about what’s happening to everybody.
Autistic Deaths and Aluminium Overload: What the Autopsies Reveal
To further support the understanding of autistic and neurodivergent vulnerability, in 2018, a team of researchers led by Professor Christopher Exley at Keele University published a landmark paper titled “Aluminium in brain tissue in autism”, in which they recorded some of the highest aluminium levels ever measured in human brain tissue -not in elderly patients or individuals with Alzheimer’s, but in young people diagnosed with autism spectrum disorder (Exley et al., 2018).
What made this finding especially significant was not just the concentration of aluminium, but its intracellular location. Aluminium was found primarily inside microglia and astrocytes -immune-related brain cells involved in neuroinflammation and detoxification. This indicates an active immune response, and raises urgent questions about vulnerability to aluminium-related injury among autistic individuals.
“The aluminium content of brain tissue in autism was consistently high. Some of the highest values for aluminium in human brain tissue were recorded” (Exley et al., 2018, p.1).
The same study proposed that autistic individuals may have an enhanced ability to recruit inflammatory cells to the brain, which in turn may carry aluminium from the bloodstream into neural tissue -a process not observed at comparable levels in neurotypical individuals. The researchers hypothesised that this might explain the elevated aluminium burden in these brains and the potential neurotoxicity arising from vaccine-derived aluminium adjuvants or other environmental exposures (Exley, 2017).
In simpler terms: autistic individuals may not be more exposed than others, but they are far more affected. Their brains are not defective -they are biologically different, and these differences include greater vulnerability to neurotoxins, oxidative stress, and inflammatory insult.
This research has been dismissed, buried, or silenced under the weight of institutional pressure. Funding for Exley’s lab was pulled shortly after this publication, and his career effectively dismantled despite decades of internationally respected work on aluminium neurotoxicity.
Importantly, the levels of aluminium observed in these brains were not observed in neurotypical control groups and that discrepancy matters. It points not to aluminium as a cause of autism -but to aluminium as a trigger for injury in already sensitive individuals, many of whom are born with folate cycle polymorphisms (such as MTHFR, MTRR) and methylation impairments that affect detoxification.
Thus, this study provides more than correlation -it offers pathological confirmation of what some may have long suspected:
Autism is not caused by vaccines -but autistic people may be injured by them if they contain toxic elements they cannot eliminate.
The real question is not “What causes autism?”
The question is: Why do we have clear pathological data showing toxic overload in autistic brains -and yet we are still refusing to adjust protocols and environments to protect them?
Until that question is answered, and until genetically susceptible individuals are removed from one-size-fits-all public health models, the system remains complicit in preventable harm.

If a significant subset of the population is genetically unable to process synthetic folic acid efficiently – and if that same group is also autistic – then mandatory folic acid fortification without genetic screening or alternative options becomes a form of, at best, biochemical discrimination and, at worst, eugenics.
9.5 Pharmacogenomic Feedback Loops and the Suppression of Neurodivergent Lineages
As previously mentioned, neurodivergent individuals – including autistic, ADHD, bipolar disorder, epilepsy, and schizophrenia – share overlapping patterns of genetic vulnerability. A growing body of research highlights the involvement of polymorphisms in genes such as MTHFR, COMT, CACNA1C, SCN2A, and BDNF, which influence neurotransmitter metabolism, methylation capacity, calcium signalling, and synaptic plasticity.
These same gene variants can modulate drug metabolism and sensitivity, making neurodivergent individuals biologically more susceptible to both environmental insults and pharmacological side effects.
The MTHFR C677T polymorphism has been strongly associated with autism, schizophrenia, and major depression (Morris et al., 2022). The COMT Val158Met variant affects dopamine clearance and has been linked to bipolar disorder and schizophrenia (Fan and Sklar, 2009). CACNA1C, which encodes a subunit of the L-type calcium channel, has been implicated in bipolar disorder, schizophrenia, ADHD, and autism (Psychiatric Genomics Consortium, 2015). SCN2A variants are highly penetrant in epilepsy and autism spectrum disorders (Wolff et al., 2017), while the BDNF Val66Met polymorphism (rs6265) has been associated with altered neuroplasticity and susceptibility to affective disorders and schizophrenia (Notaras et al., 2015).
These genetic susceptibilities converge in a clinical reality: neurodivergent populations are significantly more likely to be prescribed psychiatric or neurological medications, many of which carry teratogenic, neurotoxic, or epigenetic risks. For example, autistic individuals are up to six times more likely to experience epilepsy, with prevalence estimates ranging from 20–30% depending on intellectual disability and age (Tuchman and Rapin, 2002; Wirrell et al., 2020). Similarly, individuals with schizophrenia are up to five times more likely to develop epilepsy, with strong evidence of a bidirectional relationship and overlapping pathophysiology (Clarke et al., 2023). This clustering of genes also helps account for the high incidences of co-occurring neurodivergences within the neurodivergent population, reflecting shared pathways and intersecting biological mechanisms.
This creates a pharmacogenomic feedback loop: individuals with neurological sensitivity are diagnosed with psychiatric conditions; those conditions are treated with medications that further disrupt neurodevelopment; future generations inherit both the traits and the risks. The population most biologically reactive to pharmacological harm is also the most medicated. As a result, neurodivergent lineages are disproportionately exposed to intergenerational epigenetic risk, without informed genomic screening, mitigation strategies, or long-term tracking.
Valproate remains one of the most concerning examples. A known teratogen, valproate is associated with a 10% risk of major congenital malformations and up to a 40% risk of neurodevelopmental ‘disorders’, including autism, in exposed offspring (Veisani et al., 2021). Emerging research also points to transgenerational effects, with heritable epigenetic changes persisting beyond the directly exposed generation (Wang et al., 2022).
This dynamic is not simply a flaw in care – it is a systemic misalignment at the genomic level, where the expression of difference becomes the rationale for medical suppression. The outcome is a generational cycle of biochemical interference, in which the neurodivergent are medicated into containment rather than supported into thriving. If this feedback loop is not recognised and addressed, the most sensitive and perceptive lineages may continue to carry the burden of pharmaceutical risk -while the system that harmed them remains unexamined.
9.6 White Matter Abnormalities in Children
Evidence suggests a plausible link between impaired folate metabolism and white matter abnormalities, particularly in paediatric populations (Crider et al., 2012). The synthesis and maintenance of cerebral myelin – a key component of white matter – depends on effective methylation pathways, which require active folate (5-methyltetrahydrofolate) and vitamin B12 as cofactors. Disruption of this process, whether through genetic polymorphisms (e.g., MTHFR C677T) or accumulation of unmetabolised synthetic folic acid (UMFA), may result in functional folate deficiency, leading to compromised myelination and neurological dysfunction (Bailey & Ayling, 2009; Roffman et al., 2011). White matter lesions have been reported in children with cerebral folate deficiency syndrome, a condition associated with autoantibodies to folate receptor alpha (FRα) and are known to co-occur with motor disturbances such as ataxia, spasticity, or gait abnormalities (Ramaekers et al., 2005; Moretti et al., 2005).
Building on previously discussed research in Section 9.3 (Frye et al, 2013) on FRAAs – evidence supports the therapeutic use of 5-methyltetrahydrofolate (5-MTHF) and vitamin B12 in children presenting with white matter abnormalities and motor dysfunction, particularly in the context of cerebral folate deficiency (CFD) and impaired methylation. A 2023 case study involving two siblings with CFD reported significant improvements in developmental delays, seizure activity, and MRI-documented white matter lesions following treatment with folinic acid, later transitioned to 5-MTHF (Almahmoud, Mekki, and El Hattab, 2023). The children exhibited restored gait function, improved muscle tone, and enhanced cognitive responsiveness. Vitamin B12 has also been shown to play a crucial role in myelin synthesis and neurodevelopment; deficiencies are associated with hypotonia, delayed milestones, and coordination issues. Supplementation with B12 – particularly in conjunction with active folate – has led to clinical improvements in multiple paediatric case series, including children stated as previously ‘misdiagnosed’ with cerebral palsy or neurodevelopmental ‘disorders’ (Quadros, Sequeira and Brown, 2016).
9.7 Reframing Recovery: Misdiagnosis or Misunderstanding of Neurodivergence?
Case reports, such as those documented by Al-Mahmood, Mekki, and Al-Hattab (2023), have demonstrated that supplementation with vitamin B12 and active 5-methyltetrahydrofolate (5-MTHF) has led to significant neurological improvements in children previously diagnosed with cerebral palsy, developmental delay, or autism spectrum disorder (ASD). While these findings are promising, they raise a critical interpretive concern: are these children recovering from a misdiagnosis of a neurodevelopmental condition, or from an iatrogenic or biochemical injury that was superimposed onto a pre-existing neurodivergent identity?
The framing of such recoveries often implies that the original diagnosis was a mistake. However, this narrative fails to account for the distinction between neurodivergence and injury. A child may possess innate neurodivergent traits – such as autistic sensory processing, attention differences, or alternative communication styles – while also suffering from biochemical impairments such as cerebral folate deficiency (CFD). Treating the folate deficiency may improve seizures, motor issues, or immune dysregulation without “reversing” neurodivergence itself. To imply otherwise is to conflate recovery from injury with the erasure of identity. Perhaps neurodivergent identity has always been difficult to understand or recognise diagnostically in scientific or medical systems without additional neurological injury.
This underscores the importance of dual recognition: neurodivergent individuals can be harmed by biochemical or toxic exposures, and those harms can be mitigated – without pathologizing or mischaracterising the underlying neurotype. When the biomedical literature reports that a child was “misdiagnosed,” it often reflects a failure to differentiate between core neurobiology and secondary injury-related symptoms. This misrepresentation, however unintentional, contributes to the ongoing systemic confusion between neurodivergence and pathology.
Further evidence suggests that early intervention with methylation cofactors may reverse or halt neurological decline when white matter involvement is caught in time (Al Aref et al., 2024). These findings underscore the urgent need for early metabolic screening and individualized treatment in children exhibiting neuroimaging abnormalities and motor impairment.
Given that synthetic folic acid can inhibit folate receptor transport and mask underlying deficiency states, its role in white matter disease – particularly in vulnerable populations – warrants urgent and systematic investigation.
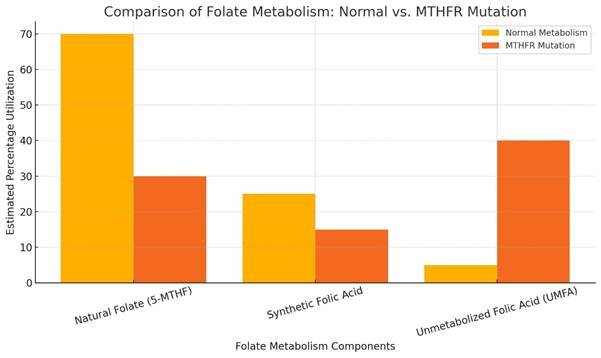
9.8 The Deceptive Framing of Autism and Intellectual Disability
Despite advances in genetic research, the dominant framing around autism and intellectual disability (ID) has dangerously shifted toward depersonalized, gene-centric explanations – most notably, the “de novo mutation” theory (Chen and Reich, 2021). While positioned as objective science, this narrative continues to evade environmental culpability and deflects away from the systemic harm that disproportionately affects autistic children, many of whom live with unacknowledged neurological damage.
A pivotal study published in Paediatrics (Maenner et al., 2023) revealed a dramatic 500% increase in autism diagnoses without intellectual disability between 2000 and 2016, compared to a 200% increase for autism with ID. This shift is stated as reflecting both improved awareness and a reclassification trend – redefining autism further away from frequent co-occurring cognitive impairments and instead towards behaviourally manageable traits.
However, this reclassification trend may obscure a deeper biological reality: the nature of neurodevelopmental harm itself may have changed. Historically, prenatal and early-life collective exposures to potent teratogens – such as thalidomide, valproate, lead, and high-dose synthetic folic acid – have been associated with more severe neurological impairments, including intellectual disability, motor dysfunction, and seizure disorders (Moore et al., 2000; Christensen et al., 2013; Vargesson, 2009). In recent decades, the intensity of these assaults has shifted, from catastrophic environmental exposure scenarios to more chronic, subclinical, but still biologically disruptive exposures, such as low-level accumulation of unmetabolised folic acid (UMFA), endocrine disruptors, and cumulative pharmaceutical interactions (Obeid et al., 2015; Troen et al., 2006).
This suggests that the declining rates of intellectual disability in autism do not necessarily reflect a “healthier” neurodevelopmental trajectory, but rather a reconfiguration of harm: the injuries are less visible but still present, manifesting as executive dysfunction, emotional dysregulation, and sensory integration challenges without overt IQ reduction. As such, rising rates of “autism without intellectual disability” may represent a population of neurodivergent individuals still affected by neurobiological injury – just in more subtle and socially tolerable forms.
In summary, this shift masks the role of environmental exposures in contributing to neurological damage, particularly in children with metabolic vulnerabilities (e.g. MTHFR variants). The study (meanner et al., 2023) identified racial and socioeconomic disparities in diagnosis, especially in the under-detection of autism without ID in black and Hispanic children, suggesting that diagnostic criteria are not being applied consistently – further reinforcing systemic bias (Maenner et al., 2023; Landrigan, 2010; Rossignol and Frye, 2014).
9.9 The Genetic Smokescreen: De Novo as Diversion
Genetic research has increasingly attributed autism–ID overlap to de novo mutations – spontaneous, non-inherited genetic changes. These mutations are often presented as random, unavoidable, and entirely genetic (Iossifov et al., 2014; Satterstrom et al., 2020). However, emerging research indicates that the rate of de novo mutations is directly influenced by environmental factors (such as prenatal drug exposure, synthetic folic acid overload, and oxidative stress), meaning these mutations are not random, but induced (Bennett et al., 2023).
Many of the genes involved in these mutations are tied to synaptic and neural function – particularly genes involved in methylation, mitochondrial performance, and neurotransmission (De Rubeis et al., 2014). However, mainstream literature consistently and repeatedly fails to explicitly connect these findings to real-world toxic exposures, creating the illusion of internal genetic failure rather than external interference.

9.10 Unfit to Parent – A Hidden Bioethical Cost
One of the most devastating consequences of poorly understood or ignored genetic vulnerabilities lies in their intersection with child protection systems. Increasingly, parents with chronic illnesses, neurological conditions, or neurodevelopmental diagnoses – many of which may be linked to folate metabolism impairments – face heightened scrutiny, legal intervention, or even loss of custody due to institutional perceptions of incompetence (Lightfoot et al., 2011; Cosey-Gay et al., 2018).
In the absence of informed public health policies, families affected by synthetic folic acid exposure and methylation dysfunction often become collateral damage. Their symptoms – fatigue, cognitive overload, emotional dysregulation – are frequently pathologized without consideration of the biochemical environment that produced them (Rossignol & Frye, 2014; James et al., 2004). When these traits appear in both parent and child, they are often misinterpreted as generational dysfunction rather than intergenerational toxicological impact (Plumptre et al., 2015).

This has led, in multiple documented cases, to the forcible removal of children from homes where genetic or epigenetic impairments were misread as signs of neglect or incapacity (Lightfoot et al., 2011). These removals are rarely scrutinized through a biomedical lens, and families are seldom offered support, genetic testing, or nutritional interventions that could stabilize health outcomes (Bailey & Ayling, 2009).
In this context, public health decisions around fortification become bioethical flashpoints. Policies that fail to account for metabolic and neuroimmune differences not only impose physiological burdens, but can also result in family separation, systemic trauma, and legal disenfranchisement (Burton & Hardaway, 2012). These outcomes extend far beyond nutrition – they represent a profound failure of informed governance.
Mandatory folic acid fortification is currently framed as a universal public health intervention; however, the socioeconomic realities of modern dietary access reveal a silent form of selection pressure – an echo of eugenics hidden in plain sight. This section explores how gluten-free access functions as a class-based filter in the modern biochemical landscape.
9.11 The Controlled Narrative: Misattributing Cause and Effect
The prevailing societal narrative usually suggests that a multitude of factors – ranging from vaccines to dietary components might “cause” autism. However, this perspective is oversimplified and misleading; a more nuanced and accurate understanding is that these factors do not “cause” autism but instead exacerbate health issues in those who are already autistic or genetically predisposed. This distinction is crucial and aligns with the genetic differences seen in autistic individuals.
The explosion of autism diagnoses in recent years has also been accompanied by a troubling misperception: that autism itself is inherently disordered. This narrative stems from a medical system that only historically recognized autistic individuals when their neurotype was paired with severe cognitive impairment or intellectual disability – disabilities that were debatably caused by unacknowledged pharmaceutical or environmental harm (Landrigan, 2010; Rossignol and Frye, 2014).
It can therefore be argued that autism in its purest form is not a disorder, it is a neurological divergence – a different operating system, and not a damaged one. A 2019 study published in Frontiers in Psychology highlights that autistic social patterns are often interpreted as deficits simply because they diverge from neurotypical expectations – not because they are inherently dysfunctional (Milton et al., 2019). When autistic people interact with each other, communication often flows more easily, suggesting that the so-called “impairments” are relational differences, not intrinsic defects.
The distinctive traits that society frequently associates with “severe autism” (e.g. cognitive impairment, seizures, intellectual disability) likely reflect those of additive injury, additional neurological damage, and not intrinsic dysfunction. These symptoms are not autism – they are collateral damage superimposed onto autistic bodies by a world saturated with toxic exposures and medical interventions; toxins that are genetically tolerated by neurotypicals.
Multiple systems and figures have previously mistaken response for identity. This narrative has pathologized the wounded neurodivergent as representative of autism itself; this is both scientifically flawed and ethically devastating. If industry and society were to remove the synthetic toxic burden – the pharmaceuticals, the food additives, the chronic immune insults – autism without interference could be reclaimed, and identified as it truly is. However, this narrative has continued throughout many decades of additional damage, and the pharmaceutical industry and its multi-generational impact on vulnerable populations would no longer be shielded by public ignorance if cause and effect were attributed appropriately.
9.12 Genetic Vulnerability, Medical Stigma, and the Weaponisation of Compliance
In the prevailing architecture of modern public health, compliance is often portrayed as a moral imperative. However, beneath this rhetoric lies a dangerous erasure: the dismissal of genetic vulnerability and the vilification of those who refuse or question medical interventions due to biological risk. This section explores how the structural ignorance of gene-environment interactions – particularly in relation to detoxification, methylation, and immune regulation – has led to a culture where medical refusal is pathologised, rather than understood.
The dominant medical model relies on a one-size-fits-all paradigm. Yet this framework collapses under the weight of mounting evidence showing that individuals with genetic variants – such as MTHFR, MTRR, COMT, CACNA1C, and BDNF – exhibit impaired processing of environmental toxins, synthetic compounds, and pharmacological agents (Bailey & Ayling, 2009; Kishi et al., 2013; Rai et al., 2020). These individuals are not rare outliers, but represent a significant subset of the population whose exclusion from medical discourse constitutes a breach of both scientific integrity and bioethical duty.
During the COVID-19 pandemic, for instance, public messaging around MRNA vaccines employed a now-familiar tactic: social compliance framed as altruism. The narrative was clear – those who refused were not only putting themselves at risk but endangering society. This rhetorical strategy, however, ignored the reality that many who declined the vaccine did so due to known adverse reactions, history of autoimmunity, or genetic vulnerability to immune dysregulation. These legitimate concerns were recoded as selfishness, recklessness, or ignorance (Simko & McCartney, 2022). The moralisation of compliance weaponised public sentiment and enabled the pharmaceutical industry to offload responsibility for harm onto the very individuals they put at risk.
Stigma, isolation, and the medical silencing of the vulnerable individuals who refused medication or vaccination on medical grounds have routinely been labelled anti-science or non-compliant, leading to job loss, exclusion from education and healthcare, and even estrangement from family. This societal shaming functions as a form of distributed enforcement, maintaining pharmaceutical dominance without the need for overt censorship. It is a form of soft eugenics, wherein those with genetic sensitivity are coerced into silence or compliance under threat of social erasure (Sullivan, 2020; Rose, 2019).

The result of this environment is a perfect loop of plausible deniability: public health agencies can claim to serve the majority, industries benefit from mass uptake of interventions, and dissenters are discredited before their warnings are even considered. Regulatory bodies, by failing to require genetic screening or to acknowledge population-specific risk, continue to uphold a system that disproportionately harms neurodivergent, immunocompromised, and epigenetically sensitive populations (García-Bailo et al., 2012; Chapman & Bilbo, 2019).
This report proposes a reframing: that medical refusal rooted in genetic and environmental awareness is not a threat to public health, but a vital act of data protection. These individuals function as sentinels – early responders to toxic overload and pharmacogenomic mismatch. Silencing them not only inflicts personal harm, but dismantles the body’s last natural warning systems. The defence of medical freedom, when informed by vulnerability, must be recognised as an evidence-based act of resistance, not fringe dissent.
Toward Inclusive Pharmacology and Epigenetic-Safe Medicine
If the medical and pharmaceutical industries are to retain legitimacy, they must move beyond reactive harm mitigation and begin actively designing treatments with genetic diversity in mind. This includes not only acknowledging mutations such as MTHFR, MTRR, and COMT, but embedding them into drug safety trials, dosage modelling, and risk assessment frameworks. The continued exclusion of epigenetically sensitive populations constitutes a form of systemic discrimination – one that withholds viable medical care from those most in need. Instead of forcing conformity through blanket protocols, future pharmacological development must embrace precision by design, creating compounds and protocols that do not harm those with impaired methylation, folate processing, or detoxification. Until this happens, a significant portion of the population will remain effectively unmedicable – not by choice, but by institutional neglect.
10. Autistic Vulnerability to Ultra-Processed Food
Ultra-processed foods (UPFs) are now strongly associated with increased risk of metabolic syndrome, gut microbiome disruption, chronic inflammation, and neurodegenerative processes (Monteiro et al., 2019; Fardet and Rock, 2021). For autistic individuals – who already face higher baseline rates of gastrointestinal issues, mitochondrial dysfunction, immune dysregulation, and genetic vulnerabilities such as MTHFR polymorphisms (Critchfield et al., 2011; Rossignol and Frye, 2014) – the cumulative impact can be devastating. In essence, the foods most likely to be tolerated by autistic individuals are also the most biologically damaging to them. This creates a cruel loop: the safer the food feels, the more harm it may do over time. Autistic people frequently rely on “safe foods” with consistent taste, texture, and predictability, which are often classified as ultra-processed (Cermak et al., 2010; Sharp et al., 2013). This dynamic is rarely discussed in public health discourse, yet it is critical to understanding the lived reality of autistic people in a toxic food system. To address rising chronic illness within neurodivergent populations, society must look beyond individual behavior and acknowledge the systemic failure to provide safe, nourishing, sensory-friendly food options.

10.1 The Gluten-Free Explosion: A Symptom of Systemic Injury
The exponential rise in gluten-free food sales (Research and Markets, 2023) is not merely a reflection of dietary trends – it is a loud, unacknowledged cry of systemic harm from synthetic folic acid. While celiac disease affects a small percentage of the population, the market data reveals a global phenomenon: people are abandoning gluten products en masse.
The global gluten-free market has more than doubled between 2022 and 2024, with projections expected to rise through 2032 (Research and Markets, 2023). This mass shift strongly correlates with increasing rates of gastrointestinal disorders and bowel-related symptoms (Sathe et al., 2022) with research suggesting that bread and wheat-based products may be triggering illness beyond medically defined gluten intolerance (rubio-Tapia et al., 2009)
Autistic individuals, those with autoimmune conditions, and genetically susceptible populations – particularly those with MTHFR polymorphisms – are often among the first to suffer from fortified or chemically altered wheat-based products (Delcoigne et al., 2020). Their likely forced migration toward gluten-free diets is not preference – it seems to be survival. The graph above illustrates this trend, validating the lived experiences of those whose bodies are rejecting what was once a true dietary staple.
Although gluten-free products are often adopted as a protective dietary measure – especially by individuals with sensitivities or genetic vulnerabilities such as MTHFR mutations – many commercially available gluten-free foods are themselves fortified with synthetic folic acid (Gluten Free Watchdog, 2021). This presents a significant and under-recognised issue, whereby individuals attempting to avoid mandated fortification may inadvertently be exposed via “health-oriented” alternatives. Ingredient labels are frequently misleading; manufacturers may list “vitamin B9” without clarifying that it refers to synthetic folic acid rather than natural folate (Bailey and Ayling, 2015) – complicating informed consumer choice and undermining medically necessary dietary modifications.
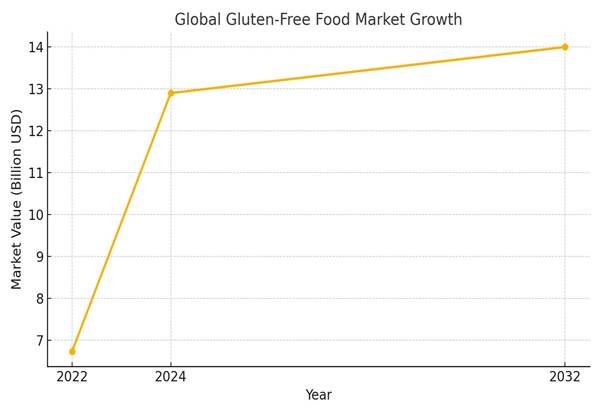
10.2 The Feedback Loop of Manufactured Intolerance
The dramatic rise in gluten-free product availability is not only a biological signal of population-level intolerance – it also reflects a strategic economic shift. Corporations that originally fortified the wheat supply and introduced synthetic additives into processed foods have now positioned themselves as leaders in the gluten-free market. This evolution highlights a closed economic loop: entities contributing to widespread dietary intolerance through product formulation now profit from its remediation.
Recent market analyses show that the global gluten-free market is projected to reach over $13 billion by 2030, driven in part by perceived health benefits and corporate diversification – not solely by the medical needs of individuals with celiac disease or gluten sensitivity (Grand View Research, 2024). Marketing efforts have capitalized on these perceptions, often overshadowing the relatively low prevalence of clinically diagnosed gluten-related disorders (BusinessWire, 2024; Extension, Oklahoma State University, 2023).
Although the gluten-free product market has expanded substantially over the past decade, access to reliable, disaggregated, country-specific consumption data remains limited. Existing reports predominantly reflect market value estimates from commercial research firms, which often conflate medically necessary gluten avoidance (e.g., Celiac Disease) with elective dietary trends, thereby distorting the interpretive value of such data. Moreover, raw metrics – such as national gluten-free prescription rates, healthcare-linked dietary exemptions, or population-level diagnosis statistics – are rarely available in the public domain. This data gap presents a substantial barrier to independent verification and hinders academic inquiry into potential correlations between gluten avoidance, environmental exposures, and policy-driven fortification programs. The persistent opacity suggests a degree of informational gatekeeping, likely influenced by commercial interests, which undermines scientific transparency in a field increasingly intersecting with public health ethics (Grand View Research, 2023).

This system exemplifies a self-reinforcing cycle: injury; sometimes reframed as disorder, is monetized through its remedy. Vulnerable populations – including autistic individuals and those with impaired metabolic pathways – are far from protected by this model; they are disproportionately affected and exploited by it. The commodification of dietary restriction thus raises ethical concerns about systemic harm being repackaged as consumer empowerment.
10.3 Coeliac Disease vs. The Gluten-Free Boom: A Critical Disparity
The global prevalence of coeliac disease is estimated to be between 0.5% and 1%. Even accounting for an average annual increase of 7.5%, (Mayo Clinic, 2023) the rise in coeliac diagnoses cannot possibly explain the $13.67 billion gluten-free market projected by 2030. If only 1% of the population had coeliac, we would expect modest niche growth in gluten-free products. Instead, we are seeing exponential market expansion – outpacing population-based medical need. This proves that the gluten-free boom is not driven by Coeliac alone – a vast, unmeasured population is experiencing symptoms from something else – modern bread is triggering illness beyond medically defined Gluten Intolerance (Rubio-Tapia, A., et al., 2009) The food system – and its synthetic additives, fortification compounds, preservatives, and altered wheat profiles – has made basic staples toxic to a growing share of the population. The data doesn’t just show a health trend – it exposes an environmental signal of harm. The gluten-free boom seems to be the market shadow of widespread biological rejection.
10.4 Diagnostic Sabotage and Data Erasure in Coeliac Testing: A Systemic Obstacle to Truth
While coeliac disease is recognised as a serious autoimmune condition, current diagnostic practices serve as a gatekeeping mechanism that limits both individual diagnosis and broader epidemiological understanding. Standard protocols – such as those endorsed by the NHS and NICE – require patients to be actively consuming gluten for several weeks before undergoing serological tests or intestinal biopsy. This creates a profound ethical conflict for individuals who experience severe reactions to gluten but are told they must harm themselves in order to access care.
Although lesser known, there are alternative diagnostic options, including HLA genetic testing, which can reliably rule out coeliac disease without requiring gluten reintroduction. These are referenced in international guidelines (NICE, 2015; Lundin & Wijmenga, 2015), yet they remain systemically withheld as part of routine care. This obstructs diagnosis for thousands – possibly millions – of individuals whose pathology falls outside the rigid boundaries of current clinical orthodoxy.
This is not just a failure of care; it is a form of diagnostic sabotage, deliberately shaping public health data by excluding anyone who does not comply with pre-set criteria. The result is data distortion on a mass scale: by refusing to diagnose those who cannot tolerate gluten product reintroduction, the system ensures that many sensitive individuals remain unofficially “non-coeliac,” thus preserving the illusion that sensitivity is rare and harmful side effects of flour fortification are minimal.
If people were en masse reporting to their doctors that they could no longer tolerate wheat, and were subsequently tested negative for coeliac – then that would be telling in itself. This diagnostic gatekeeping means the majority of people suffering symptoms will not get tested for celiac and will remain a hidden statistic.
This also has knock-on effects on research funding, policy development, and public perception. If the data reflects only a narrow diagnostic window, then gluten-related disorders appear statistically insignificant. This gives cover to ongoing fortification policies and dismisses growing anecdotal and clinical evidence that gluten – or additions to the flour-may be neuroinflammatory or immunogenic for a far broader subset of the population – especially those with gene mutations like MTHFR, MTRR, and COL1A1, who already have reduced detoxification capacity.
This structural concealment represents not just epistemic injustice but active manipulation of scientific and medical truth.
Through erasing the population most harmed by gluten, regulators maintain a policy framework that protects industry interests while leaving vulnerable populations invisible, unsupported, and medically gaslit.
10.5 The Cost of Survival: Socioeconomic Barriers to Wellness
While the gluten-free boom may appear to reflect increased awareness or improved dietary choices, there is a deeper and more disturbing reality beneath it – one of economic gatekeeping. Gluten-free food is consistently priced at approximately twice the cost of its wheat-based counterparts (Lee et al., 2007). For neurodivergent individuals – especially autistic people – this cost is not about lifestyle, but potentially survival.
Many genetically vulnerable individuals who react adversely to fortified bread, processed wheat, and synthetic additives are effectively forced onto gluten-free diets to manage chronic symptoms. For those in lower socioeconomic brackets, access to gluten-free options is severely limited (Pohl, Tzianetas and Reilly, 2021).
This creates a bioeconomic divide where only those with financial means can consistently access the foods their bodies tolerate. In this reality, food is no longer just sustenance – it becomes a filter. A mechanism by which health, cognition, and quality of life are selectively preserved or degraded along class lines (Huh and Gordon, 2020). For autistic people, this means that wellness becomes a privilege, not a right. The ones who remain healthy are often those who can afford to circumvent the toxins. The rest risk being left behind – medicated misdiagnosed, and further pathologised; this is not equity, this is quiet eugenics of access, masked as market freedom (Berg and Pineda, 2020).
Many foundational studies on folic acid fortification fail to adequately represent populations with known genetic susceptibilities, including individuals with MTHFR, MTRR, and DHFR variants. These polymorphisms significantly affect folate metabolism and may lead to increased accumulation of unmetabolised folic acid (UMFA), reduced methylation capacity, and heightened vulnerability to oxidative stress and inflammation.
Despite increasing awareness of genetic variants -most public health research does not stratify outcomes by genotype. As a result, the aggregated data may obscure disproportionate harm to genetically susceptible individuals, particularly within neurodivergent, chronically ill, and racially marginalized groups.
This omission not only weakens the scientific validity of fortification claims but also raises serious ethical questions about equity in public health policy. Without disaggregated data, at-risk populations remain invisible in the evidence base, excluded from meaningful risk-benefit analysis and denied access to informed consent.
Policies built on such omissions risk perpetuating harm under the guise of public good -an oversight that must be addressed to restore both scientific integrity and ethical accountability.
11. Recent Health Trends Post Folic Acid Fortification

Historically, the UK did not require folic acid fortification in bread (unlike many countries, including the USA – who has been mandated since 1998). However, in September 2021 the UK government announced plans to mandate the addition of folic acid to non- whole meal wheat flour to prevent neural tube defects. The legislation was introduced in November 2024, with implementation expected by the end of 2026 (UK GOV, 2024).
However, In the United Kingdom, food manufacturers were permitted to fortify products with synthetic folic acid long before any national mandate was introduced. This led to widespread, unregulated exposure without informed consent across the population, especially through breakfast cereals, spreads, and dietary supplements.
By 2006, the Scientific Advisory Committee on Nutrition (SACN) warned that mandatory fortification could push certain groups above the tolerable upper intake level unless voluntary fortification was controlled (British Dietetic Association, 2023). Similarly, in Ireland, voluntary industry fortification led to a 30% increase in folic acid intake among women of childbearing age, influencing delays in national policy due to concerns of overexposure (Sayers et al., 2009). These cases highlight how the fortification rollout was, in effect, already underway – quietly, without public scrutiny or clear oversight.
While this policy is promoted as a public health measure, growing evidence contradicts its supposed benefits and highlights serious health risks associated with synthetic folic acid fortification.
Synthetic folic acid has been linked to:
- Increased colorectal cancer risk – Studies have shown a significant rise in colorectal cancer cases following fortification policies (Vollset et al., 2013).
- Immune suppression & autoimmune disorders – High levels of UMFA have been associated with reduced natural killer (NK) cell activity, making the immune system less effective in fighting infections and cancer cells (MDPI, 2023).
- Metabolic disorders & epigenetic Risks – Excessive folic acid intake interferes with one-carbon metabolism, affecting DNA methylation, gene expression, and long-term metabolic health (Ondicova et al., 2022).
- Neurodevelopmental disabilities – UMFA accumulation during pregnancy and infancy raises concerns about disrupted methylation pathways, potential cognitive effects, and increased risk of neurological damage (Bailey et al, 2010).
11.1 The Rising Tide of Early-Onset Colorectal Cancer
Recent studies have revealed a significant and alarming increase in colorectal cancer (CRC) among younger adults. This trend is particularly evident in both the United States and the United Kingdom, where incidence rates have been rising for decades – a shift that cannot be fully explained by improved screening alone. The American Cancer Society (American Cancer Society 2024) estimates 152,810 new CRC cases in 2024, with 53,010 anticipated deaths – a slight increase from the previous year. Notably, while overall CRC rates have declined due to improved screening and lifestyle changes, individuals under 50 have experienced a 2.4% annual increase in CRC incidence from 2012 to 2021. This trend has prompted health authorities to adjust screening guidelines, recommending earlier and more frequent screenings to address the escalating risk in younger populations.
- In the United States, individuals aged 20 to 29 experienced a 7.9% annual increase in Colorectal Cancer cases between 2004 and 2016, while those aged 30 to 39 saw a 4.9% annual rise in the same period (Vuik et al., 2019).
- Further research by Siegal et al., reveals a further increasing rise (Siegel at al., 2019).
- Similarly, in England, colorectal cancer incidence among adults aged 20 to 29 rose from 0.8 per 100,000 in 1993 to 2.8 per 100,000 in 2014, reflecting an average annual increase of 8% (Araghi et al., 2019).
- Further research by Cancer Research UK reveals a further increasing rise (Cancer Research UK, 2020).
Colorectal cancer, historically considered a disease of older populations, is now striking younger individuals at an accelerating rate (Vuik et al., 2019; Siegal et al., 2019; Cancer Research Uk, 2020). This rise coincides with major environmental and dietary shifts, including the introduction of folic acid fortification in food supplies. Given the known biochemical interactions between synthetic folic acid, unmetabolised folic acid (UMFA), and cellular methylation processes, this factor warrants urgent investigation.
The prevailing explanation that screening advancements alone account for the rise does not hold up under scrutiny (Ahnen et al., 2014). If this were the case, we would expect to see an initial spike in cases, followed by a plateau or decline – as screening identifies previously undiagnosed cancers and then stabilizes. Instead, the steady and continuous rise in early-onset colorectal cancer suggests a genuine increase in disease incidence, not just detection bias.
Colorectal cancer (CRC) incidence among individuals under 50 in the United States, particularly around the 1998 folic acid fortification, reveals notable trends:
- Research has highlighted a concerning rise in rectal cancer among younger adults, which cannot be explained by improved screening alone. In the United States, between 2000 and 2013, rectal cancer incidence increased by 3.2% annually in individuals aged 20 to 29 (Siegel et al., 2017).
- Meanwhile, in the United Kingdom, rectal cancer incidence among adults aged 20 to 39 increased by 4.4% annually between 1990 and 2014 (Araghi et al., 2019). These findings align with global trends, signaling a shift toward earlier-onset colorectal cancer that demands further investigation into environmental, dietary, and biochemical influences.
What makes this shift especially concerning is its timing – closely aligned with the introduction and expansion of mandatory folic acid fortification policies. While folic acid was introduced under the premise of preventing neural tube defects, research suggests that excessive intake, especially in those with impaired folate metabolism (such as individuals with MTHFR mutations), may disrupt cellular methylation processes and increase cancer risk. These biochemical interactions – specifically the accumulation of unmetabolised folic acid (UMFA) – raise concerns regarding long-term health outcomes.
Given the timeline, factors such as synthetic folic acid fortification and unmetabolised folic acid (UMFA) accumulation warrant serious scrutiny as potential contributors to this unprecedented rise.
11.2 Prostate Cancer Incidence: Rising Trends
A 2014 meta-analysis by Tio et al., titled Folate Intake and the Risk of Prostate Cancer: A Systematic Review and Meta-Analysis challenges conventional wisdom regarding folate’s role in cancer prevention. While natural folate has long been associated with positive health outcomes, this study raises serious concerns about the effects of elevated serum folate levels – particularly in relation to prostate cancer risk.
The findings from Tio et al’s study (2014) suggest a clear distinction between dietary folate from natural food sources and the synthetic folic acid commonly used in supplementation and food fortification. While dietary folate does not appear to increase prostate cancer risk, elevated serum folate levels were correlated with a significantly higher incidence of prostate cancer.
This distinction is critical, as fortified foods and synthetic folic acid supplementation artificially raise serum folate levels, bypassing the body’s natural regulatory mechanisms. The Tio et al., 2014 study highlights the following concerns:
Increased Serum Folate and Cancer Risk:
- The meta-analysis found that higher circulating folate levels were linked to increased Prostate cancer incidence. This aligns with previous studies suggesting that excessive Folic Acid supplementation may fuel tumour growth rather than provide protection (Tio et al., 2014).
Given that folic acid fortification is mandatory in many countries, the unintended consequences of elevated serum folate levels must be critically examined. The assumption that “more folate is always better” is not supported by the data and is likely putting specific populations at a highly increased risk.
United Kingdom:
Prostate cancer has long been considered a disease of older men, with most cases diagnosed in individuals over 65. However, recent data indicates a significant and concerning increase in prostate cancer incidence among younger men, particularly in the United Kingdom.
- Prostate cancer is now the most common cancer among men in the UK, accounting for 28% of all male cancer cases between 2017 and 2019 (Cancer Research UK, 2023).
- Notably, since the early 1990s, prostate cancer incidence in men aged 25 to 49 has increased by a staggering 622%. This sharp rise raises serious concerns about environmental and dietary factors contributing to disease onset beyond traditional risk factors like age and family history (Cancer Research UK, 2023).
The timeline of this rise correlates closely with the early voluntary introduction of synthetic folic acid into the UK food supply, well before mandatory fortification. As the UK’s Scientific Advisory Committee on Nutrition (SACN) noted in their 2006 report:
“There is evidence that the average intake of (synthetic) folic acid in the UK has increased over time, partly due to the voluntary fortification of some foods such as breakfast cereals, and the increased use of supplements.” (Scientific Advisory Committee on Nutrition, 2006, p.39)
This voluntary fortification began as early as the 1980s and expanded rapidly during the 1990s – precisely the same timeframe in which prostate cancer incidence among men aged 25–49 began to climb dramatically. Although current policy bodies do not acknowledge a causal link, the coincidence between early-life exposure to unmetabolised folic acid and delayed-onset hormone-sensitive cancers like prostate cancer must be urgently reconsidered. Dismissing such correlations as “unproven” is no longer scientifically or ethically defensible.
United States:
- Prostate cancer remains the most diagnosed cancer among men in the U.S., with an estimated 313,780 new cases expected in 2025 (Zerocancer.org, 2025).
- While still rare in younger men, incidence rates among those aged 45 to 54 have begun rising in recent years (CDC, 2024).
The overall incidence rate stands at 116.5 per 100,000 men annually, reinforcing the disease’s significant impact (National Cancer Institute, 2023).
Prostate cancer remains the most diagnosed cancer among men in the U.S., with an estimated 313,780 new cases expected in 2025 (Zerocancer.org, 2025). While most cases still occur in older men, incidence rates among younger men aged 45 to 54 have begun rising in recent years, signaling a shift in age distribution (CDC, 2024).
A frequent counterargument in epidemiology posits that rising prostate cancer rates reflect improved screening technologies, particularly the introduction of Prostate-Specific Antigen (PSA) testing in the early 1990s. However, this explanation fails to fully account for the observed patterns. If screening alone were responsible, we would expect a temporary spike in diagnoses followed by a plateau or decline – yet prostate cancer rates have continued to rise steadily, even as screening practices remained relatively stable (Rynecki et al., 2023).
This persistent increase suggests a real rise in disease incidence, not merely detection bias. One underexplored variable is the introduction of synthetic folic acid fortification, which became mandatory in the U.S. food supply in 1998 (Oakley, 1999). Since then, population-wide exposure to unmetabolized folic acid (UMFA) has become near-universal. Emerging evidence links UMFA accumulation to disrupted one-carbon metabolism, hormone-sensitive cancers, and immune dysregulation (Troen et al., 2006; Vollset et al., 2013; Crider et al., 2012).
Given the biological plausibility and temporal overlap, further investigation is urgently needed to explore whether folic acid fortification may be contributing to the rise in prostate cancer – particularly in younger age groups.
11.3 The Bigger Picture: Unmetabolised Folic Acid and Cancer Growth
The 2014 meta-analysis (Tio et, al, 2014) study revealed that while dietary folate intake alone was not significantly associated with prostate cancer, elevated blood folate levels were linked to a statistically significant increase in risk – suggesting that synthetic folic acid exposure and UMFA accumulation, rather than natural folate, may be driving this association (Tio et al., 2014).
A growing body of evidence suggesting that synthetic folic acid does not behave like natural folate in the body and that unmetabolised folic acid (UMFA) has been detected in high concentrations in individuals consuming fortified foods and supplements (Pfeiffer et al., 2015), raising concerns about its potential role in cancer progression, immune dysfunction, and methylation interference.
If a subset of the population – especially those with genetic polymorphisms – are disproportionately affected by folic acid fortification, then public health policies must be reconsidered. Failing to do so risks turning well-intended fortification programs into a silent contributor to rising cancer rates, especially for vulnerable populations.
12. Foundational Studies -The Teratogen Switch. Investigating the Methods
Fortification Influencing Studies: Canada
In 1949, Paradione (paramethadione), a highly teratogenic anti-epileptic drug, was marketed in Canada and known to cause severe birth defects including facial malformations and neural abnormalities (U.S. FDA, 2004; German, Kowal and Ehlers, 1970; Zackai, Mellman and Neiderer, 1975). Though there is no official Health Canada statement confirming its market withdrawal, multiple pharmacological records and drug compendiums ceased listing the drug after 1994 (Merck Index, 1996), suggesting its discreet removal. This withdrawal coincided with the early phase of Canada’s folic acid fortification trials and likely introduced a confounding variable that has not been accounted for in key public health data.
In 2007, the pivotal study by De Wals et al. claimed a very significant 46% reduction in neural tube defects (NTDs) in Canada following folic acid fortification between 1993 and 1997 (De Wals et al., 2007). However, this timeframe overlaps with the suspected withdrawal of paramethadione. Notably, there were later recalls of both Epival (brand name of divalproex sodium/ Depakote) and valproate in 2007 –with the pharmaceutical industry officially claiming “errors in formulation warranting a recall – and the need to seek an ongoing alternative medication in future” (Health Canada, 2007).
These removals represent significant pharmaceutical interventions capable of skewing data in public health trends and NTD incidence.
The De Wals et al., study (2007) failed to account for the removal of teratogenic drugs during its observation window. Without controlling for this variable, improvements in NTD rates were misattributed solely to folic acid fortification. Crucially, the study was not independent. It was led by Health Canada’s Food Directorate -the very agency that mandated folic acid fortification – creating a direct conflict of interest. The study also involved the British Columbia Ministry of Health, which enforces fortification policy, and the Robert Stempel College of Public Health (Miami, FL), alongside multiple publicly funded universities. These institutions had a clear vested interest in producing positive outcomes to validate their own mandates.
This timing of the removal of all three teratogens does not appear to be a mere coincidence.
By delaying the removal of valproate and Epival (Canadian brand name) until 2007 (Health Canada, 2007), regulators conveniently avoided a sharp, suspicious drop in data of birth defects immediately following paramethadione’s quiet withdrawal. Instead, staggering the removals across more than a decade engineered continuity – this would ensure that the decline in NTDs appeared gradual, organic, and easily attributed to fortification. This appears to be a disturbing cover-up by design;
if all three teratogens had been withdrawn at once, the data would have shown a far more dramatic and questionable shift in data.
Scientific Misconduct Through Omission
The study by De Wals et al. (2007), often cited as a cornerstone of folic acid fortification policy, attributed the decline in neural tube defects (NTDs) in Canada to mandatory folic acid fortification. However, critical analysis reveals that this conclusion was drawn without accounting for a major confounding factor: the potential withdrawal of paramethadione, a highly teratogenic anti-epileptic drug, in 1994.
Multiple medical textbooks and pharmacovigilance reports ceased listing paramethadione as available in Canada after 1994. This introduces a significant confounding variable that would have greatly reduced NTD incidence, irrespective of folic acid fortification.
Unlike the US., Health Canada has no publicly accessible documentation on the paramethadione withdrawal –even though it was once marketed there. Absence of accessible public record, combined with its disappearance from pharmaceutical listings in 1994, strongly suggest a covert withdrawal strategy –potentially due to mounting concerns over teratogenicity.
12.1 Critique of De Wals et al. (2007) Study Using STROBE and Scientific Standards
The study by De Wals et al. (2007), which attributed a significant reduction in neural tube defects (NTDs) to folic acid fortification in Canada, fails to meet several key scientific and ethical standards for observational research. Most notably, it omits essential confounding factors – such as the contemporaneous withdrawal of teratogenic anti-epileptic drugs (AEDs) – thereby violating multiple elements of the STROBE guidelines (Von Elm et al., 2007).
1. Failure to Account for Confounding Variables
STROBE item 7 requires researchers to “clearly define all potential confounders” and specify how they were addressed in the analysis. De Wals et al. (2007) did not mention or control for the withdrawal of known teratogens such as paramethadione and mephenytoin from the Canadian market during the pre-fortification era – factors which could significantly alter baseline NTD rates independent of folic acid exposure. This omission undermines the validity of their causal claims.
2. Inadequate Justification for Causal Attribution
Although ecological studies can suggest associations, STROBE item 20 emphasizes that limitations must be explicitly discussed, especially when making causal inferences. De Wals et al. present folic acid fortification as the definitive cause of NTD reduction, despite the lack of control groups and the presence of alternative explanations (e.g., drug regulation, diagnostic coding changes). The authors’ failure to address these limitations violates the principle of scientific neutrality and overextends the data’s inferential power.
3. Ethical Oversight and Transparency
According to the Tri-Council Policy Statement (CIHR, NSERC & SSHRC, 2018), Canadian research involving human data must disclose known influences and competing causal mechanisms. De Wals et al.’s omission of pharmaceutical policy changes during the relevant timeframe may constitute a breach of ethical transparency, particularly given the study’s public health implications.
4. Editorial Responsibility and Peer Review
The New England Journal of Medicine adheres to the International Committee of Medical Journal Editors (ICMJE) standards, which require that conclusions be “firmly grounded in evidence” and proportionate to the data presented (ICMJE, 2019). The publication of a strong causal claim without adequate consideration of confounding events raises concerns about the robustness of the peer review process in this instance.
However, critical variables were not accounted for. Notably, the authors failed to disclose the concurrent withdrawal of multiple teratogenic anti-epileptic drugs, such as Paradione (paramethadione), from the Canadian market between 1994 and 1998. The omission of this data – despite its temporal overlap with the purported success of fortification -represents a serious confounder that would have significantly skewed outcome attribution.
Furthermore, the De Wals et al. (2007) study does not account for changes in fertility trends, prenatal screening access and abortion data, or diagnostic reporting practices during the same period. Without these controls, causal claims linking fortification directly to reduced NTD rates remain speculative at best.
This failure to adjust for confounding variables violates core principles of epidemiological study design, particularly when making population-level causal inferences. Moreover, such omissions suggest a pattern of selective framing that aligns more closely with policy justification than with scientific integrity.
12.2 FDA Archive
During this investigation, the author retrieved an archived FDA webpage (screenshots and contents on the next page) referencing the drug paramethadione, specifically noting its approval in 1949 and its final discontinuation order dated as 1989 (Author’s screenshot 1,2 and 3, 2025; Annexe A, 2025). This archived page is no longer publicly listed or directly accessible through standard FDA databases and was located via a broken external reference link (FDA, N.D) – highlighting the fragility and selective accessibility of regulatory data across digital timelines.
(Author’s screenshot 1, 2025)
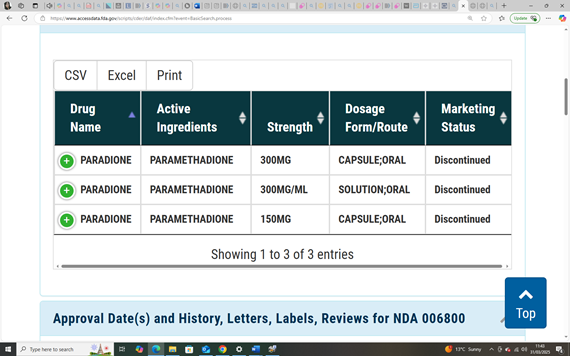
(Author’s screenshot 2, 2025)
(Author’s screenshot 3, 2025)
Authors screenshot 4 (Captured March 2025)
Annexe A – Archived FDA Record (Captured March 2025)
U.S. Food and Drug Administration
Search
Search FDA
Submit search
Drugs@FDA: FDA-Approved Drugs
New Drug Application (NDA): 006800
Company: ABBVIE
Products on NDA 006800
CSVExcelPrint
| Drug Name | Active Ingredients | Strength | Dosage Form/Route | Marketing Status | TE Code | RLD | RS |
| PARADIONE | PARAMETHADIONE | 300MG | CAPSULE;ORAL | Discontinued | None | No | No |
| PARADIONE | PARAMETHADIONE | 300MG/ML | SOLUTION;ORAL | Discontinued | None | No | No |
| PARADIONE | PARAMETHADIONE | 150MG | CAPSULE;ORAL | Discontinued | None | No | No |
Showing 1 to 3 of 3 entries
Approval Date(s) and History, Letters, Labels, Reviews for NDA 006800
Original Approvals or Tentative Approvals
CSVExcelPrint
| 06/15/1949 | ORIG-1 | Approval | Label is not available on this site. |
Showing 1 to 1 of 1 entries
Supplements
CSVExcelPrint
| 03/24/1989 | SUPPL-15 | Manufacturing (CMC) | Label is not available on this site. | |
| 12/02/1987 | SUPPL-14 | Manufacturing (CMC) | Label is not available on this site. | |
| 12/07/1984 | SUPPL-13 | Manufacturing (CMC)-Expiration Date | Label is not available on this site. | |
| 06/28/1982 | SUPPL-12 | Manufacturing (CMC)-Control | Label is not available on this site. | |
| 07/14/1980 | SUPPL-10 | Labeling | Label is not available on this site. | |
| 10/24/1979 | SUPPL-8 | Manufacturing (CMC)-Control | Label is not available on this site. | |
| 07/29/1975 | SUPPL-5 | Manufacturing (CMC)-Control | Label is not available on this site. |
Despite the drug’s clear discontinuation timeline, and its well-documented teratogenic risks by the 1970’s (German, Kowal and Ehlers, 1970; Zackai, Mellman and Nedeirer, 1975), no mention of paramethadione appears in CDC literature or public-facing communications about declining neural tube defect (NTD) rates in the early 1990s (CDC, 2024). Instead, the reduction in NTDs has been attributed exclusively to the implementation of folic acid fortification.
Notably, the record lists AbbVie Inc. as the manufacturer and 1975 as the first manufacture date. This presents a chronological discrepancy: AbbVie was not established until 2013, following a corporate spin-off from Abbott Laboratories – the known original patent holder of paramethadione (FDA, 2004; United States Patent and Trademark Office, 2011).
The presence of AbbVie’s name on a regulatory record dated decades before the company’s formation (Author’s screenshot 1,2 and 3, 2025; Annexe A) suggests that regulatory databases may have been updated post hoc, reflecting modern corporate structures rather than preserving accurate historical manufacturer data. This has broader implications for transparency and accountability in long-term pharmacovigilance.
The erasure of early manufacturing records and the post hoc appearance of paramethadione on FDA databases as originating in the 1970s (Author’s screenshot 1,2 and 3, 2025; Annexe A) – despite its original approval in 1949 by Abbott Laboratories – raises serious questions about regulatory compliance and historical transparency.
Under the 1962 Kefauver-Harris Amendments to the U.S. Food, Drug, and Cosmetic Act, all pre-1962 drugs were required to undergo retrospective efficacy review via the Drug Efficacy Study Implementation (DESI) process (U.S. Congress, 1962; FDA, 2013). The apparent absence of this audit trail for paramethadione, coupled with its quiet reappearance under institutional successors, may represent a breach of both federal regulatory standards and public disclosure obligations.
If manufacturing entries were retroactively inserted or manipulated to exclude the drug’s original teratogenic history, this constitutes a potential falsification of federal records. Moreover, the quiet continuity between Abbott’s original patent and AbbVie’s later regulatory footprint -despite AbbVie not existing at the time -suggests not only institutional inheritance of benefit but a deliberate bypass of liability. In this context, the disappearance of documentation should not be seen as an oversight, but as a signal: an intentional disruption of the historical record to obscure corporate culpability and regulatory complicity.
12.3 Boxed Insight: Evidence of Digital Obfuscation in Public Drug Records
While investigating the regulatory trail of paramethadione – a known teratogenic anti-epileptic drug originally patented by Abbott Laboratories and quietly transitioned to AbbVie – anomalous digital behaviour emerged. A download link referencing “drug ban: paramethadione” from a U.S. government regulatory archive did not lead to pharmaceutical or safety documentation as expected. Instead, it opened an Excel spreadsheet containing a Federal Register entry titled:
“HISA Anti-Doping and Medication Control Rule, Notice, Federal Trade Commission” (Citation: 88 FR 5070, Date: 2023)
This document details regulatory protocols under the Horse Racing Integrity and Safety Act of 2020, which is unrelated to human pharmaceutical oversight. The file, purportedly matching a banned teratogenic human drug, instead discusses anti-doping guidelines for equine sport.
Implications:
- The mismatch suggests data masking or redirect substitution: a tactic often used to bury or misdirect sensitive information.
- The themed crossover – “medication control” and “anti-doping” – may be intentional camouflage, using a regulatory document with parallel keywords to obscure access to the true record.
- Combined with earlier evidence of absent recall logs and litigation sealing orders around paramethadione, this incident adds further weight to claims of systemic obfuscation and digital censorship.
This finding underscores the importance of nonlinear investigative methods in exposing how pharmaceutical data is filtered, redirected, or altogether hidden from public scrutiny -particularly when that data could expose industry malpractice or governmental complicity.
Such structural gaps in the regulatory record may contribute to the erasure of key contributors to declining neural tube defect rates, particularly if drug withdrawals – like that of paramethadione – are excluded from public health narratives in favour of a singular causal framework.
12.4 A Pattern of Protection: Regulatory Collusion Across Generations
When viewed in isolation, the FDA’s delay in updating valproate pregnancy warnings and the missing regulatory trail of paramethadione may appear as separate failures – one a modern regulatory stall, the other a historical omission; but when placed side by side, these events form a disturbing continuum. What we are witnessing is not a series of disconnected missteps, but a multi-decade pattern of institutional protection – where Abbott Laboratories and its corporate offshoot, AbbVie, are repeatedly shielded from full accountability by the very regulatory body meant to safeguard the public.
Examining two Cases:
As stated, in the case of paramethadione, Abbott’s 1949 teratogenic drug, there are no clear records of its withdrawal, no DESI audit under the 1962 Kefauver-Harris Amendments, and no public-facing explanation for its disappearance. Yet, in the 1970s, the FDA began listing the drug again under restructured documentation – despite AbbVie not even existing at that time. This ghost record re-entry appears post hoc, a paper trail rewritten after the fact to potentially sever liability and preserve institutional control. The FDA did not correct the missing data; it preserved the absence.
Decades later, with valproate (Depakote), Abbott submitted formal requests to update pregnancy safety labels in 2005 and 2007. These were blocked by the FDA, in closed-door discussions, despite escalating evidence of neural tube defects and cognitive impairment in exposed infants. Once again, critical warnings were withheld from the public, and the delay – later leveraged to absolve corporate responsibility in court – contributed directly to avoidable harm. It was only in 2011, after the damage had peaked and the policy benefit extracted, that warnings were officially amended.
Both cases demonstrate the same architecture of complicity:
- A known teratogen.
- A suppressed or erased regulatory record.
- A corporate entity shielded from scrutiny.
- And an FDA that facilitated silence rather than enforcing truth.
The alignment between regulatory delay and manufactured policy success—particularly in the use of NTD data to justify folic acid fortification—mirrors the timeline of suppressed paramethadione data. This is not coincidence. This is historical repetition, strategic forgetting, and collaborative deception.
What emerges is a clear narrative:
Abbott and AbbVie were protected. The FDA was complicit. The public paid the price.
This is not the story of a single drug. It is the story of a system that learned how to bury one teratogen – and then repeated the same play, decades later, with a more sophisticated mask.
12.5 Undocumented Exposure: The Ghost Variable in the China–US Trial
Valproate, as previously mentioned is a known teratogen strongly associated with neural tube defects (NTDs), especially spina bifida (Jentink et al., 2010). In China, the drug is referred to as 丙戊酸钠 (sodium valproate) and was eventually approved under the brand name 德巴金 (Dé bā jīn) – but not until 2001, years after the China-US Collaborative Project on neural tube defects prevention had concluded (PHIRDA, 2021).
However, population pharmacokinetic studies confirm that valproate was already in clinical use in Chinese hospitals as early as 1993, implying unofficial access prior to formal market approval (Wang et al., 2015). This raises an alarming possibility: that valproate was being distributed or prescribed without transparent oversight during the critical trial window (1993–1995), only to be formally recognized once its contribution to birth defect rates could be politically and scientifically dismissed.
If valproate usage was reduced, replaced, or quietly withdrawn during the same period that folic acid supplementation was being tested, the NTD reduction attributed to folic acid may, in fact, be due to a pharmaceutical variable deliberately excluded from analysis. The delayed regulatory recognition of valproate in China not only raises ethical concerns but also suggests a deliberate obfuscation of causality -a pharmaceutical shadow that haunts the foundation of global fortification policy.
The China-US Collaborative Project on neural tube defects Prevention is heralded as definitive proof that folic acid prevents birth defects. However, that critical piece of the puzzle was never disclosed: the role of valproate, a widely used antiepileptic drug known to cause neural tube defects like spina bifida (Jentink et al., 2010).
Evidence shows valproate was being used in Chinese hospitals before its formal approval in 2001 under the brand name Dé bā jīn (PHIRDA, 2021). Clinical pharmacokinetic studies conducted in China as early as 1993 confirm that sodium valproate was administered to paediatric epilepsy patients during this period (Wang et al., 2015), meaning it was in circulation during the exact timeframe of the China–US folic acid trial (1993–1995) (Berry et al., 1999).
If valproate – either branded as Epilim or in generic form – was quietly restricted, substituted, or withdrawn during the same period folic acid was being introduced, the observed reduction in neural tube defects would have been falsely attributed to the folic acid. The study did not control for medication exposures, nor did it acknowledge the teratogenic risk posed by drugs in use at the time.
This omission represents a critical failure in the study’s design; nutritional science was instead, pharmaceutical silence – and a global mandate was built on that omission.
12.6 The MRC Vitamin Study
The MRC Vitamin Study (MRC, 1991) is perhaps the most controlled – but also very misleading. While it found that high-dose folic acid (4 mg/day) reduced NTD recurrence in high-risk women, it did so under heavily filtered conditions. Critically, the MRC Vitamin study (MRC, 1991) excluded women with epilepsy and therefore eliminated pharmaceutical teratogens like valproate from the pool entirely (Québec E&M Working Group, 2019). It also administered high-dose synthetic folic acid (4 mg/day) to women who were potentially free from MTHFR polymorphisms, polypharmacy, or toxic metabolic burden. These were not metabolically or environmentally representative subjects -they were, in effect, the ideal responders. No data was gathered on unmetabolised folic acid (UMFA) accumulation, gene–nutrient interactions, or long-term outcomes.
12.7 The Fourth Pillar: Policy Without Proof
The fourth most cited “study” supporting folic acid fortification is not a study at all -it is the 1992 CDC policy recommendation that all women of childbearing age consume 400 micrograms of synthetic folic acid daily (CDC, 1992). This directive was not drawn from new empirical data, nor did it test folic acid supplementation in a broad or representative population. Instead, it extrapolated findings from the previously mentioned MRC Vitamin Study (MRC, 1991) – a trial that involved a high-risk but highly controlled cohort, specifically women who had previously experienced an NTD-affected pregnancy.
Yet it was this narrow snapshot that became the foundation of a globally expanding policy. The CDC codified it, other governments followed, and folic acid fortification became a global default -without ever testing the intervention across a genetically diverse, pharmaceutically burdened, or environmentally vulnerable population.
Beyond discrediting all three foundational studies, this report section presents evidence of institutional misconduct and strong indicators of unlawful and unethical concealment tied to folic acid fortification policy.
13. Influence and Warning Regarding Fortification
13.1 Figures: The Oakley Influence
One of the most influential figures in shaping the global folic acid fortification agenda was Dr. Godfrey P. Oakley, a former CDC official that could be referred to as the “father of folic acid fortification” (Oakley, 2002). Over several decades, Oakley authored or co-authored more than 400 publications advocating for mandatory fortification across dozens of countries, frequently framing any resistance to fortification as a public health failure.
While Oakley’s contributions to neural tube defect (NTD) prevention are historically significant, his ongoing influence in global policy discussions has raised questions about scientific impartiality and rhetorical dominance. Many of his most cited publications rely on unstratified population data, often omit discussion of genetic polymorphisms affecting folate metabolism, and advocate for population-wide interventions over personalized medicine (Oakley, 1997; Crider, Bailey and Berry, 2011).
Oakley was also a vocal opponent of genetic screening in relation to folic acid metabolism, explicitly arguing that such measures were unnecessary and that universal fortification would suffice (Oakley, 1997). This stance – while well-intentioned – undermined calls for a precision-nutrition approach at a time when evidence of metabolic diversity was already emerging.
His influence extended not only through direct authorship, but through repeated citation by regulatory bodies, public health reviews, and global health initiatives. Oakley’s work forms the backbone of many fortification endorsements, including those referenced in the Cochrane systematic review that continues to shape international policy (De-Regil et al., 2015).
The continued prominence of a single, ideologically committed figure in policy discourse underscores a deeper concern: when scientific consensus becomes tethered to personality rather than methodological diversity, policy risks becoming resistant to new data. In this case, it has contributed to a self-reinforcing narrative that elevates early findings while downplaying the complexity of emerging genetic and biochemical evidence.
13.2 The Contradictions: Data That Didn’t Fit the Narrative
While early fortification studies were widely promoted as evidence of success, subsequent data sets have emerged that challenge the simplicity of that narrative. Several countries that implemented mandatory folic acid fortification failed to observe sustained reductions in neural tube defect (NTD) rates, and in some cases, experienced plateaus or reversals in trend.
For instance, in both the United States and Australia, public health surveillance has noted that NTD rates began to stabilize – or in certain demographics, increase – despite ongoing fortification and widespread supplementation (Mai et al., 2019; Riley et al., 2021). These findings suggest that the initial declines were likely due to other variables – such as improved prenatal diagnostics, targeted education campaigns, or the phaseout of teratogenic pharmaceuticals – rather than fortification alone.
Moreover, studies that attempted to stratify data by ethnicity or socioeconomic status have revealed disparities in outcomes, with some vulnerable populations exhibiting little benefit or even potential harm associated with synthetic folic acid exposure (Tinker et al., 2010). These complexities were often excluded from policy summaries or relegated to footnotes, leaving the public-facing narrative largely unchanged.
The continued citation of early, unstratified success stories – while omitting or minimizing newer contradictory evidence – represents a form of scientific cherry-picking that undermines the credibility of the public health messaging around folic acid. For a policy intervention with such far-reaching implications, a higher standard of evidence, transparency, and adaptability is essential.
13.3 Unmetabolised Folic Acid: Population-Level Saturation, Metabolic Bottlenecks, and Foetal Exposure
The safety of synthetic folic acid has been repeatedly questioned by researchers in both Ireland and the United Kingdom, particularly regarding its metabolism, bioaccumulation, and transplacental passage. A growing body of evidence now indicates that the presence of unmetabolised folic acid (UMFA) is not an isolated phenomenon, but a widespread biological reality with concerning implications for long-term health – especially in genetically or developmentally vulnerable populations.
A pivotal study by Sweeney et al. (2009) evaluated circulating folate forms in healthy adults living in Ireland under conditions of voluntary folic acid fortification. The researchers found that over 80% of individuals had detectable levels of UMFA, despite the absence of a mandatory fortification policy. This suggested a baseline saturation of the folate pathway, even at relatively modest exposure levels, and raised important questions about the long-term consequences of chronic low-level folic acid intake across the general population (Sweeney et al., 2009).
To understand why UMFA accumulates, the Wright, Dainty, and Finglas (2007) study explored folic acid metabolism in healthy adults. The researchers identified that human dihydrofolate reductase (DHFR) activity is both limited and variable, resulting in a metabolic bottleneck that prevents complete conversion of synthetic folic acid to biologically active forms. This mechanistic insight confirmed that even moderate supplemental intake can exceed enzymatic capacity, leading to unmetabolised folic acid circulating in the bloodstream (Wright, Dainty and Finglas, 2007).
Building on these findings, McNulty et al. (2013) investigated folate levels in pregnant women and umbilical cord blood. The study revealed that UMFA was not only present in maternal serum, but also crossed the placenta and accumulated in foetal circulation, even in a country without mandatory fortification. This confirmed that developing foetuses are being exposed to synthetic folic acid in utero, with potential implications for neurodevelopment, immune programming, and long-term health outcomes (McNulty et al., 2013).
Together, these studies form a critical triad:
- Wright et al. (2007) explained the metabolic limitations that lead to that persistence;
- Sweeney et al. (2009) showed that UMFA persists at the population level;
- McNulty et al. (2013) proved that foetal exposure is occurring.
These findings raise fundamental concerns regarding current fortification policy frameworks and supplement guidelines, particularly in light of genetic polymorphisms such as MTHFR, which further impair folate metabolism and exacerbate the risk of UMFA-related toxicity in vulnerable populations.
13.4 Emerging Risk of Early-Onset Cancer from Unmetabolised Folic Acid Exposure
Research by Sweeney et al. (2009) and Wright et al. (2007) confirms that unmetabolised folic acid (UMFA) persists in the general population due to widespread synthetic intake and metabolic saturation. McNulty et al. (2013) provides clear evidence that UMFA crosses the placenta, resulting in foetal exposure during critical periods of neurodevelopment. These findings carry grave implications – not just for theoretical risk, but for the tangible reality of early-life health outcomes. When this exposure continues after birth – via infant formula, fortified foods, and paediatric supplements – it embeds a cumulative epigenetic signature during the most vulnerable stages of biological programming. Studies such as Kim (2004) and Lucock et al. (2021) have shown that disruptions in folate metabolism and methylation are linked to cancer development. In this context, early and repeated UMFA exposure may not just increase long-term cancer risk – it may bring forward onset into childhood. Given the compounded effects across prenatal and postnatal environments, especially in genetically vulnerable populations, it is not alarmist but medically responsible to raise concern that we may soon see a rise in paediatric cancers driven by cumulative folate pathway disruption. Public health narratives have yet to confront this emerging crisis, but the signals are
These warnings were very clear by 2013, yet over twelve years later mandatory folic acid fortification has been aggressively expanded worldwide – despite mounting evidence of its serious long-term accumulation and health risks.

13.5 Policy Conditions and Research Freedom: Ireland’s Unique Position
Ireland’s voluntary approach to folic acid fortification during the 2000s created a rare opportunity for scientific inquiry prior to full-scale policy implementation. Unlike the United States, which mandated folic acid fortification in 1998, Ireland maintained a more flexible system, allowing researchers to observe real-world folate exposure and its metabolic consequences under non-compulsory conditions. This policy window enabled pivotal studies – such as Sweeney et al. (2009), which documented persistent unmetabolised folic acid (UMFA) in over 80% of adults, and McNulty et al. (2013), which identified UMFA in both maternal and neonatal cord blood. These findings emerged in a context where national policy was still under debate, giving Irish researchers the freedom to critically assess biological risks without the constraints often imposed by entrenched regulatory frameworks or pharmaceutical lobbying. In contrast, countries with long-standing mandatory programs, such as the United States and Canada, have seen limited inquiry into UMFA’s potential harm, likely due in part to policy entrenchment and the political cost of questioning established public health interventions (Sweeney et al., 2009; McNulty et al., 2013; Berry et al., 2010).
13.6 A Mass Genetic Risk Framed as a Fringe Detail
MTHFR polymorphisms are not rare exceptions – they are prevalent across multiple populations (Moll and Varga, 2015). Individuals with MTHFR mutations cannot convert synthetic folic acid into its active form efficiently (Scaglione and Panzavolta, 2014). As a result, UMFA builds up in their systems (Bailey and Ayling, 2009) while functional folate remains deficient at the cellular level (Stover, 2004).
This paradox leads to impaired methylation, DNA repair dysfunction, poor detoxification, and heightened susceptibility to neurodevelopmental Disorders, chronic illness, and cancer (Troen et al., 2006; Pfeiffer et al., 2015; Sweeney et al., 2009). Yet public health agencies ignore this widespread vulnerability because acknowledging it would invalidate the blanket safety narrative surrounding fortification.
The omission of MTHFR isn’t just a scientific oversight – it is strategic. It shields policy from accountability by suppressing variables that would demand complexity, screening, and nuance. This neglect is policy armor; it allows institutions to continue mandating folic acid fortification while claiming it is universally beneficial.
In doing this, it betrays the populations most at risk – those with the very genetic vulnerabilities that synthetic folic acid was never designed to accommodate.
This report does what those studies did not; it names the gene. It traces the metabolic consequences, and it demands that MTHFR – and those who carry it – are brought back into the conversation because when public health erases the very biology that makes people vulnerable, it stops being science altogether
13.7 Controlled Opposition? Suppressed Warnings: Independent Researchers
While much of the literature on folic acid fortification has been shaped by institutional allegiance and rhetorical minimization, a handful of independent voices raised serious concerns. They asked questions and, in many cases, they were ignored or overshadowed by the more politically palatable narrative of universal fortification.
The Health Council of the Netherlands (Health Council of The Netherlands, 2000)
- In their report ‘Risks of folic acid Fortification,’ the Health Council warned against unmonitored public exposure to synthetic folic acid. They emphasized the danger of masking vitamin B12 deficiency and called for further evidence before embracing national fortification. Despite this strong position, their recommendations were largely bypassed in the broader European policy agenda.
Tufts University Study on Colorectal Cancer (Mason et al., 2007)
- Researchers observed a correlation between mandatory folic Acid fortification and increased colorectal cancer incidence. They suggested this could be a result of folic acid accelerating the growth of existing neoplasms. Although framed with caution, the implications were clear – and dismissed by policymakers.
Is Folic Acid Good for Everyone? (Smith, Kim and Refsum, 2008)
- Published in the American Journal of Medicine, this paper raised a direct question about whether universal folic acid supplementation was suitable for all populations. The authors emphasized risks to subgroups with metabolic vulnerabilities and called for personalized approaches. Their message, while calm in tone, was a clear warning: blanket policies may harm the very populations they aim to protect.
Despite affecting an estimated 30–60% of various global populations (Moll and Varga, 2015) -with notable ethnic variations- the MTHFR mutation is almost never addressed in public health documents, scientific reviews, or policymaking discourse related to folic acid fortification. This silence does not appear accidental; it is apparently systemic.
13.8 Respectable, Informed, and Contained: The Authors Who Stayed Within the Lines
The authors of the 2008 paper ‘Is Folic Acid good for everyone?‘ – A. David Smith, Young-In Kim, and Helga Refsum – are not fringe figures. They are well-credentialed scientists with deep expertise in folate metabolism, cancer biology, and nutritional epidemiology. Their contributions have added nuance to the discourse, and it is plausible that they were sincerely attempting to raise concerns about the blanket application of folic acid policy.
However, their critiques stop just short of systemic challenge. Key concepts like MTHFR polymorphisms, population-wide methylation disruption, and the risks of unmetabolised folic acid are consistently omitted. Whether this is due to personal caution, editorial constraint, or institutional pressure is unclear – but the result is the same: the core risk remains obscured.
The 2022 Lancet Global Health commentary, co-authored by Smith and Refsum among others, continues this pattern. It presents itself as balanced inquiry into mandatory fortification but avoids discussion of genetic variability and conflicts of interest. Smith, for example, has declared consulting ties to Aprofol AG – a company that stands to benefit from folate-related product development. These connections do not automatically invalidate the authors’ work, but they must be considered in evaluating the completeness of their critique.
It is possible that these scientists are warning within the limits available to them. It is also possible they serve a controlled opposition function -permitted to question the surface while leaving the core structure untouched. Either way, their position remains one of institutional alignment. Their caution, though informed, is carefully framed and that framing to date has shaped policy more than the evidence has.
13.9 Unacknowledged Harms
While the benefits of folic acid fortification have been widely promoted, the potential harms – particularly among genetically susceptible populations -have received disproportionately little attention in scientific discourse or policy evaluation.
Emerging evidence has linked excessive synthetic folic acid intake and unmetabolised folic acid (UMFA) accumulation to a range of adverse outcomes, including:
- Increased risk of cancer proliferation in individuals with pre-existing neoplasms (Kim, 2004);
- Suppression of immune function, including natural killer cell activity (Troen et al., 2006);
- Neurodevelopmental disruption, particularly in foetuses and infants with impaired folate metabolism (Bailey & Ayling, 2009);
- Adverse pregnancy outcomes, including higher risk of ‘autism spectrum disorder’ (additional neurological harm) in genetically predisposed children (Li et al., 2020; Schmidt et al., 2011).
Despite the availability of this data, risk-benefit analyses have remained generalized and static – rarely incorporating stratified evaluations based on genotype, cumulative exposure, or long-term health trajectories. The populations most likely to experience adverse effects -those with methylation impairments, chronic inflammation, or pre-existing neurological conditions – have largely been excluded from safety reviews.
This absence of risk stratification does not reflect a lack of scientific knowledge. Rather, it points to a systemic unwillingness to adapt public health policy in light of emerging complexity. In effect, certain harms have been institutionally normalized through omission – a form of passive harm that persists beneath the surface of official narratives.
14. Folic Acid and Epigenetic Disruption: Uncovering the Neural Impact
14.1 Altering DNA methylation
A groundbreaking 2022 study titled “Folic acid intervention during pregnancy alters DNA methylation, affecting neural target genes through two distinct mechanisms” (Ondicova et al., 2022) revealed that synthetic folic acid is far from passive in the developing foetus. Instead, it acts as a powerful epigenetic modifier, with the ability to reshape gene expression in ways that may have lasting neurological consequences.
The researchers (Ondicova et al., 2022) found that folic acid affects neural development by altering DNA methylation – a biological process that switches genes on or off. This happens via two primary mechanisms:
Direct Methylation Pathway:
- Folic acid acts as a methyl donor, directly contributing methyl groups to DNA (Ondicova et al., 2022). This process can silence or activate specific genes, including those involved in brain development, synapse formation, and neural signaling. In excess, this can lead to over-methylation of genes that should remain active, potentially impairing normal neurological pathways.
Modulation of the Methylation Machinery:
- Beyond directly donating methyl groups, folic acid also influences the enzymes responsible for DNA methylation – such as DNA methyltransferases (Ondicova et al., 2022). This alters the epigenetic landscape more broadly, potentially leading to aberrant gene regulation across multiple neural systems.
While folic acid is often promoted as a protective measure against neural tube defects (NTDs), this study (Ondicova et al; 2022) provides compelling evidence that too much folic acid – or its presence in the wrong form – can be harmful to neural development. The issue becomes even more critical in individuals with MTHFR gene mutations, who are unable to process Folic Acid effectively and are more prone to accumulating unmetabolized folic acid (UMFA). This can lead to epigenetic disruptions in utero, increasing the risk of neurological damage.
This research challenges the “more is better” approach to folic acid supplementation. It also calls into question the safety of mandatory folic acid fortification – especially in the absence of genetic screening or personalized nutrition.
Key concerns raised by this revelational study (Ondicova et al., 2022) include:
- The potential for long-term neurological effects due to altered gene expression during foetal development.
- The need for individualized approaches to prenatal nutrition, especially for genetically vulnerable populations.
- The importance of distinguishing between synthetic folic acid and natural, bioavailable folate (5-MTHF) in public health policy (Ondicova et al., 2022).
14.2 Genetic Manipulation: From Mitochondrial Gene Editing to Mass Biochemical Targeting
In October 2023, a study published in Nature Plants (Forner et al., 2023) detailed the successful targeted knockout of a conserved mitochondrial gene in plants using genome editing tools such as TALEs (Transcription Activator-Like Effectors). This deliberate and precise disruption of plant mitochondrial gene expression demonstrates a powerful, evolving toolkit in biotechnology – one capable of altering vital biological processes at the organelle level. The goal in this case was control: specific phenotypic changes in plants achieved by manipulating the organelles responsible for energy production and inherited traits.
In both plants and people, mitochondrial function and epigenetic expression are malleable (Wallace and Fan, 2010; Matzke and Mosher, 2014). In plants, it’s targeted through molecular tools. In humans, it’s being altered en masse through synthetic biochemical exposure – particularly in prenatal and early developmental windows. The plant study highlights the capability to control life at the mitochondrial level.
14.3 Folic Acid as a Genetic Disrupter
An important 2022 study published in Cancers (Basel) (Zsigra et al., 2022) provides compelling evidence that synthetic folic acid is not biologically inert – it actively alters gene expression and DNA methylation patterns in colorectal cancer cells. This means that folic acid can function as an epigenetic modifier, not just a nutrient. Researchers observed that folic acid exposure caused both hypermethylation and hypomethylation of genes involved in tumour growth, cell division, and DNA repair- precisely the processes known to be disrupted in cancer.
Individuals with impaired folate metabolism – such as those carrying MTHFR gene mutations – are less able to convert synthetic folic acid into its active, usable form. This leads to an accumulation of unmetabolised folic acid (UMFA) in the bloodstream, which interferes with DNA methylation and histone modification processes crucial to gene regulation. Methylation governs whether tumour-suppressing and tumour-promoting (oncogenic) genes are activated or silenced (Troen et al., 2006; Pfeiffer et al., 2015; Sweeney et al., 2009).
When UMFA accumulates in genetically susceptible individuals, it disrupts this critical regulatory system. Oncogenic genes may be switched on, while tumour-suppressor genes are downregulated (Bailey and Ayling, 2009; Zhao et al., 2023; Zsigra et al., 2022). These disruptions do not merely alter metabolism – they may contribute directly to cancer development, especially in individuals with MTHFR polymorphisms (Cole et al., 2007; Crider, Bailey and Berry, 2011; Troen et al., 2006).
A pivotal study by Zsigra et al. (2022) found that in HT-29 colorectal tumour cells, folic acid exposure increased methylation activity and cell proliferation – early indicators of cancer. By contrast, SW480 cells with lower methylation sensitivity showed less response. This differential impact highlights how genetic factors dictate risk, making one-size-fits-all folic acid policies ethically and scientifically untenable.
Further evidence from Tomita et al. (2013) emphasized that individuals with MTHFR polymorphisms are at greater risk of cancer when exposed to high folic acid levels. Despite clear findings, policies have failed to adjust – raising concerns of biochemical discrimination through state-sanctioned nutrient exposure.
Meanwhile, excess UMFA under fortification regimes or high-dose supplementation may competitively inhibit folate receptors and disrupt intracellular folate balance (Bailey and Ayling, 2009; Obeid et al., 2015). This can paradoxically induce a state of “functional folate deficiency” – even in the presence of high circulating folate – by impairing the utilisation of biologically necessary forms. The result is a cascade of epigenetic instability, including aberrant DNA methylation and histone modifications (Crider et al., 2012), altered immune responses (Lucock et al., 2013), and increased susceptibility to chronic disease and neurodevelopmental disorders (Waterland and Jirtle, 2003; McKay et al., 2002).
These intergenerational epigenetic effects are not theoretical – they represent a silent but profound driver of modern disease trends, demanding a reassessment of fortification policies through the lens of nutrigenomic diversity.
When a known risk is systematically imposed on a subset of the population without informed consent – particularly when that group disproportionately includes autistic, neurodivergent, or genetically nonconforming individuals – the line between public health policy and eugenics becomes dangerously blurred. The repetition of harm in the face of clear evidence can no longer be dismissed as oversight. It reflects a deeper institutional willingness to sacrifice the few for the perceived benefit of the many.
14.4 Ancient Lineages, Modern Vulnerabilities: The Genetic Heritage Ignored by Policy
The higher prevalence of MTHFR polymorphisms – particularly the C677T variant – in Hispanic and Amerindian populations is more than just a genetic curiosity. It reflects ancestral genetic signatures shaped over thousands of years by environments and diets uniquely adapted to these populations. Pre-colonial indigenous diets in Mesoamerica and South America were naturally rich in bioavailable dietary folate (Bailey et al., 2009; Lucock et al., 2022). These populations, therefore, had little evolutionary pressure to efficiently metabolise synthetic forms of folate, which were nonexistent in their ancient environments.
Colonial disruption drastically altered these traditional dietary practices, imposing nutrient-poor staples and later, fortified foods containing synthetic folic acid (Selhub & Rosenberg, 2016). Populations carrying ancient genetic adaptations found themselves abruptly exposed to a metabolic burden their biology had never encountered – leading to widespread unmetabolised folic acid (UMFA) accumulation, methylation dysfunction, and increased risk of chronic illness (Smith et al., 2008; Refsum et al., 2022).
Ignoring these ancient genetic legacies in public health policy represents not only medical negligence but a continuation of biochemical colonialism. It forces genetically distinct populations into metabolic paradigms developed without consideration for their ancestral heritage – resulting in disproportionate harm that has remained invisible in policy discourse.
15. Eugenics, Bioengineering, or Genocide?
The lines between these categories blur dangerously. Folic acid fortification, when applied across genetically diverse populations without regard for metabolic capacity, functions as a biochemical filter – silently erasing out or harming individuals based on hidden vulnerabilities (Crider et al., 2011; Safi et al., 2012). If the adverse outcomes are known (as mounting evidence suggests), and yet policies continue or expand, this transcends negligence and borders on:
- Bioengineering: Manipulating biological systems in the name of public health without informed consent and accounting for genetic diversity or adverse outcomes (Pearson, 2005).
- Eugenics: Implementing policies that disproportionately harm genetically distinct subgroups, particularly those already marginalized (e.g., neurodivergent populations and/ or those with genetic differences) (Ulrich and Potter, 2006).
- Genocide (soft form): If systemic harm and potential population reduction occur through a deliberate failure to acknowledge or address these risks (Wisconsin Medical Journal, 2023).

15.1 Eugenics and the Law
When public health policy disproportionately harms a genetically distinct subset of the population – especially when that harm is foreseeable, avoidable, and driven by systemic neglect or profit – it crosses a critical ethical and legal boundary (Ulrich and Potter, 2006; Crider et al., 2011). Eugenic outcomes, even when unintended, cannot be excused when the risks are clearly documented, ignored, and imposed on vulnerable individuals without consent or alternative.
The decision to implement mass synthetic folic acid fortification without accounting for genetic variability raises not only scientific questions, but profound ethical ones. When a public health intervention disproportionately harms certain populations – particularly those with known metabolic vulnerabilities -it ceases to be a neutral policy. It becomes a form of bioengineered discrimination, whether intentional or not.
This raises the spectre of soft eugenics: the subtle, systemic shaping of population health outcomes through chemical and nutritional interventions that ignore genetic diversity. When privileging certain metabolic pathways and silencing evidence of harm in others, such policies risk exacerbating existing health disparities under the guise of universal benefit.
More critically, these interventions are often implemented without informed consent. Genetically susceptible individuals – including those with MTHFR, MTRR, and DHFR mutations – have had no say in the biochemical regimes imposed on them through food supply manipulation. In most jurisdictions, these populations are not even acknowledged in policy documentation, much less protected.
However, discriminatory health policy or willful exposure to harm is prohibited under several key legal frameworks:
- The Human Rights Act 1998 (UK) – protects individuals from inhumane or degrading treatment and upholds the right to life and bodily integrity.
- European Convention on Human Rights (1950) – notably Article 2 (Right to Life) and Article 3 (Freedom from Torture and Inhuman or Degrading Treatment), both of which may be violated by policies that knowingly endanger genetically vulnerable groups.
- UN Convention on the Rights of Persons with Disabilities (CRPD 2006) – mandates equal protection from harm, access to informed healthcare, and the right to bodily autonomy, specifically protecting individuals with disabilities from discriminatory practices in public health.
The absence of stratified oversight echoes broader patterns of structural neglect, where those most vulnerable to harm are rendered statistically invisible. It is not enough to measure fortification’s “success” in terms of average birth defect reductions while overlooking the chronic, neurological, and immunological consequences for those on the margins of that data.
Public health policy, when rooted in incomplete evidence and implemented without accountability, can become an instrument of harm. Whether by design or default, the result is the same: long-term biomedical experimentation on populations unable to opt out.
15.2 Changing Times and Attitudes on Eugenics?
The academic article entitled “Can ‘Eugenics‘be Defended? ” (Veit et al., 2021) aims to rehabilitate the term ‘eugenics’ by distinguishing coercive, state-led historical practices from what the authors call ‘liberal’ or ‘voluntary’ eugenics. The core arguments include:
- The term ‘eugenics’ has become overly politicized.
- Genetic enhancement via technologies like CRISPR should be morally evaluated on outcomes, not semantics.
- Voluntary genetic selection could reduce suffering and increase well-being.
- Society should not avoid using gene editing for enhancement if it’s technologically feasible (Veit et al., 2021).
While framed as philosophical neutrality, the article dangerously minimizes the structural, ethical, and historical implications of eugenics. Key points of concern include:
- Ethical language is weaponized to rebrand eugenics, shifting public perception toward normalization.
- ‘Voluntary choice’ in reproductive technologies is an illusion when societal pressures, policies, and inequality shape those decisions.
- Framing enhancement as a moral obligation invites the creation of an idealized human blueprint – this is soft eugenics by design.
- There is no recognition of how ableism, classism, and bio-capitalism intertwine to define whose genes are worth enhancing.
- Recasting genetic selection as ‘beneficence’ erases centuries of harm and opens the door to futuristic population engineering.
This article represents a disturbing rhetorical pivot: the attempt to sanitize and legitimize eugenics under the guise of individual freedom, technological progress, and utilitarian ethics. It reframes control as care, and selection as autonomy. This is not philosophical inquiry – it is narrative engineering. The fact that respected academics are making this argument openly is not just dangerous – it confirms that the ideological infrastructure of eugenics has never disappeared; it has evolved.
15.3 Racial Disparities and Genetic Targeting
The systematic omission of genetic variations like MTHFR polymorphisms from public health and nutritional policies raises profound questions about bioethics, identity, and the very definition of being human. Genetics is not merely biochemical; it is an ancestral legacy carrying encoded resilience, adaptation strategies, and cultural knowledge (Jablonka & Lamb, 2014; TallBear, 2013). Ignoring this diversity – imposing biochemical uniformity through universal supplementation and fortification – is effectively a subtle form of genetic engineering. It prioritizes conformity over genetic uniqueness, risking the inadvertent reshaping of humanity’s genetic landscape over successive generations (Rose, 2007; Comfort, 2014).
Synthetic folic acid fortification is often promoted as a universal public health solution, but it fails to account for population-specific genetic differences in folate metabolism. Polymorphisms in MTHFR, MTRR, and DHFR – enzymes critical to methylation and folate bioavailability – do not occur uniformly across global populations. Studies have shown that individuals of Hispanic, Indigenous American, African, and South Asian descent may have higher frequencies of these polymorphisms compared to populations of Northern European ancestry (Botto & Yang, 2000; Bailey & Ayling, 2009; James et al., 2004).
This uneven genetic landscape means that synthetic folic acid fortification may place certain racial and ethnic groups at disproportionate biochemical risk, particularly when policies are applied without metabolic screening or dietary flexibility. For example, UMFA accumulation has been found to be more prevalent in Black, Indigenous, and Latino populations – groups that also face systemic barriers to diagnosis, detoxification support, and culturally competent care (Pfeiffer et al., 2015; Plumptre et al., 2015).
These disparities are further compounded by structural racism in medicine, where symptoms linked to methylation dysfunction or neurological inflammation are frequently dismissed, misdiagnosed, or pathologised. In some cases, neurodivergent traits in children of colour are interpreted not as signs of biomedical vulnerability, but as indicators of behavioural disorder – resulting in higher rates of school exclusion, psychiatric medication, or family separation (Burton & Hardaway, 2012; Cosey-Gay et al., 2018).
The systematic erasure of the MTHFR gene mutation from public health policy has profound biochemical and social consequences. Ignoring this polymorphism exposes millions of genetically vulnerable individuals – particularly those of Hispanic, Amerindian, Mediterranean, and East Asian descent – to chronic metabolic stress, impaired methylation cycles, and accumulation of unmetabolised folic acid (UMFA) (Bailey & Ayling, 2009; Selhub & Rosenberg, 2016). This biochemical overload does not remain contained within the current generation; it is epigenetically passed down, magnifying health disparities across generations through disrupted methylation patterns and compromised DNA repair mechanisms (Lucocket al., 2022).
Despite these compounding factors, there has been no large-scale investigation into how racialized populations may differentially experience harm from synthetic folic acid fortification. The absence of such inquiry reflects a dangerous assumption of biological homogeneity – and risks replicating colonial logics in modern biomedical policy.
Through omission of diversity, current public health paradigms implicitly establish a preferred biological identity. Those whose genetics deviate from this defined norm experience both medical marginalization and cultural erasure, challenging their fundamental right to biological integrity (Marks, 2017). As these practices persist unchallenged, they slowly redefine what it means to be human; stripping away genetic differences under the guise of public good, ultimately altering humanity itself on an epigenetic scale (Lappé & Landecker, 2015).
15.4 Eugenics by Neglect
When biomedical interventions are deployed across entire populations without accounting for genetic variability, long-term harm can occur – not necessarily through overt malice, but through structural indifference. In the case of synthetic folic acid fortification, vulnerable groups have been left unprotected by policies that presume biological uniformity, despite decades of data indicating otherwise (Botto & Yang, 2000; James et al., 2004; Bailey & Ayling, 2009).
This failure to acknowledge population-level differences in folate metabolism – particularly among those with MTHFR, MTRR, or DHFR variants – has allowed certain groups to bear a disproportionate burden of chronic illness, neurological dysfunction, and immunological impairment. And yet, because these effects manifest slowly and unevenly, they are rarely linked back to the root intervention.
The result is a form of eugenics by neglect: a public health approach that, while not explicitly discriminatory, systematically overlooks the needs of those whose biology does not conform to dominant models. The harm is not always immediate, but it is cumulative, intergenerational, and often invisible in aggregate data.
This silent bias is exacerbated by institutional resistance to adaptation. Even as evidence mounts regarding genetic vulnerability, policymakers have largely upheld the status quo – failing to update guidelines, issue warnings, or explore safer, more bioavailable alternatives like methylfolate or folinic acid. In doing so, they perpetuate a model of biomedical governance that privileges statistical averages over individualized safety.
True equity in public health demands more than universal policies – it requires precision, accountability, and the willingness to confront scientific discomfort. When systems fail to meet that standard, the ethical line between protection and control begins to blur.
15.5 Globalization of Harm – The Exportation of a Failed Model
Despite growing evidence of biological variability and unintended harms, synthetic folic acid fortification has been aggressively exported as a universal public health solution – championed by international health organizations, NGOs, and research groups with longstanding ties to the pharmaceutical and food industries. What began as a regional intervention in North America has evolved into a globalized policy model, often implemented without population-specific safety assessments (Crider et al., 2011; De-Regil et al., 2015).
As of 2024, more than 80 countries have adopted mandatory folic acid fortification programs (Crider et al., 2011). These rollouts are often justified by data from countries like Canada and the United States – despite the presence of confounding variables such as teratogenic drug withdrawals that went unreported in key studies (Ray et al., 2002). The assumption that fortification will yield uniform benefits across ethnically and genetically diverse populations remains scientifically unverified, particularly in regions with elevated prevalence of MTHFR, MTRR, and DHFR variants (Plumptre et al., 2015; Pfeiffer et al., 2015).
Low-income populations, in both the global North and South, face the greatest barriers to opting out of fortified foods. In many regions, processed foods are the most accessible calorie sources, leaving vulnerable communities with little nutritional agency or access to alternative folate forms. Without routine metabolic screening or public education about folate polymorphisms, the burden of UMFA accumulation and its downstream effects falls disproportionately on populations that are least resourced to respond (Pfeiffer et al., 2015).
This policy trajectory mirrors historical patterns of biomedical colonialism, in which Western health models are exported with minimal consideration for local ecologies, dietary patterns, or genetic diversity (Hardon and Moyer, 2014; Biehl and Petryna, 2013). Framed as ethical imperatives, these interventions often reproduce systems of control and dependence under the banner of care.
In this context, synthetic folic acid fortification – deployed as a one-size-fits-all solution without accounting for biological and cultural difference – risks becoming a biopolitical mechanism of global harm, rather than a safeguard for health.
15.6 The Illusion of Consensus
The widespread perception that folic acid fortification is a universally safe and effective intervention rests not on comprehensive, stratified data – but on a repeating loop of selective citation and institutional endorsement. Key studies, such as those by Berry et al. (1999), Crider et al. (2011), and Williams et al. (2015), are cited extensively in both scientific literature and policy documents, often without acknowledging their methodological limitations or contextual blind spots.
These publications, while influential, frequently rely on data drawn from early-stage interventions, use aggregated population averages, and fail to control for confounding factors such as drug withdrawals, prenatal screening advances, and shifting socioeconomic variables (Berry et al., 1999; Crider et al., 2011). Despite these limitations, they continue to be referenced as definitive evidence – creating a self-reinforcing system in which perceived consensus is maintained through repetition rather than rigor.
This circularity is compounded by the influence of prominent individuals and advocacy groups who have played dual roles as researchers, policy advisors, and public health spokespeople. The result is a knowledge ecosystem vulnerable to echo chambers, in which dissenting data – particularly concerning genetic vulnerability and biochemical risk – is marginalized or omitted.
The illusion of consensus is further amplified through global health organizations that replicate these foundational studies across continents without revalidating them in the context of local genotypes, health systems, or food ecologies (De-Regil et al., 2015). In doing so, what began as a precautionary intervention becomes a fixed doctrine, resistant to change – even in the face of new biochemical, epidemiological, and ethical evidence.
True consensus in science must be dynamic, capable of evolving with emerging data. When policy solidifies prematurely – anchored to outdated or incomplete evidence – it ceases to protect and instead begins to constrain.
16. Outlook: Hindsight into Foresight
16.1 Manufacturing Crisis, Marketing Cure: Cancer Incidence, Vaccines, and the Ethical Risks of Pharmaceutical Dependency
The sudden and steady rise in cancer incidence over recent decades has intersected suspiciously with the rapid emergence and aggressive marketing of cancer-preventative vaccines, particularly MRNA-based solutions (Sahin & Türeci, 2021; Moderna, 2023). Growing evidence suggests that environmental and dietary factors, including prolonged exposure to synthetic folic acid and unmetabolised folic acid (UMFA), may accelerate the proliferation of existing tumours, creating conditions conducive to increased cancer rates (Kim, 2008; Mason et al., 2007; Selhub & Rosenberg, 2016).
This raises troubling questions: Could the rise in cancer be, at least partially, a manufactured crisis – resulting from widespread biochemical exposure to substances whose long-term risks were systematically ignored or minimized? If so, the pharmaceutical industry’s readiness with preventative cancer vaccines – products that simultaneously promise public health salvation and unprecedented profits – cannot be dismissed as mere coincidence (Angell, 2005; Goldacre, 2012).
Pharmaceutical companies have historically profited from treatments for conditions partly driven by biochemical and environmental exposures that remain inadequately regulated. This creates cycles of dependency: populations harmed by one set of pharmaceutical or dietary interventions become dependent upon another set of pharmaceutical products to correct or mitigate that harm; Abbott and AbbVie are exemplary of this cycle of harm. The result is not improved health but increased pharmaceutical dependency, biological commodification, and the erosion of bodily autonomy (Rose, 2007; Lappé & Landecker, 2015).
The ethical implications are clear; failing to address the root biochemical exposures that contribute to cancer incidence, public health policy and pharmaceutical interests have converged to create a market ripe for exploitation. Unless critically examined and ethically challenged, these dynamic risks transforming public health into perpetual biological dependency.
16.2 Industrialising Urgency: The Rapid Global Rollout of Cancer Vaccine Production
The rapid scale-up and deployment of cancer vaccine production infrastructure globally raise significant concerns, particularly given the urgency generated by increasing cancer rates. BioNTech, the creator of one of the widely used COVID-19 MRNA vaccines, has established modular ‘BioNTainer’ production facilities, such as those recently inaugurated in Rwanda, capable of decentralized mass manufacturing of MRNA-based cancer treatments (BioNTech, 2023). Concurrently, Moderna, in collaboration with pharmaceutical giant Merck, employs artificial intelligence to expedite the production of personalized MRNA cancer vaccines, promising turnaround times as short as 48 hours from diagnosis to delivery (Investors, 2023).
Additionally, biotech startups such as Cellares are developing advanced automation systems like the ‘Cell Shuttle,’ capable of producing tens of thousands of individualised cell-based cancer treatments per year (Financial Times, 2023). This accelerated industrial capacity aligns suspiciously well with projected increases in cancer incidence, potentially driven by biochemical exposures like synthetic folic acid, raising ethical and safety concerns about the rush to widespread deployment without comprehensive long-term studies.
This scenario reflects historical patterns of manufactured urgency, where crises are leveraged to justify rapid, large-scale interventions before rigorous examination of long-term impacts is possible. The implications extend beyond immediate health outcomes, potentially fostering biochemical dependency, limiting regulatory oversight, and normalising invasive medical interventions on a global scale.
16.3 The Ultimate Beneficiaries: Pharmaceutical Dominance, Financial Interests, and Global Health Commodification
The rapid global rollout and industrial-scale production of cancer vaccines primarily benefits major pharmaceutical companies, biotechnology corporations, and their financial backers. Key beneficiaries include Moderna and its strategic partner Merck, companies that have heavily invested in MRNA vaccine technologies and cancer immunotherapy (Investors, 2023). BioNTech, alongside Pfizer, is also positioned to significantly profit from their established global distribution networks and advanced modular production platforms like the BioNTainers (BioNTech, 2023).
Biotechnology startups such as Cellares, supported by substantial venture capital investments, similarly stand to gain financially through the commercialization of automated production technologies designed for rapid, mass-scale personalised cancer treatment (Financial Times, 2023). This scenario highlights the role of investment groups and venture capitalists who strategically finance these biotechnology enterprises to maximize returns during health crises.
Beyond direct financial gains, these entities significantly expand their influence over global health policy and medical guidelines, further entrenching regulatory capture and eroding independent oversight (Angell, 2005; Goldacre, 2012). The resulting biochemical dependency and normalization of invasive medical interventions exemplify the growing commodification of human biology, where health becomes increasingly controlled and monetized by pharmaceutical interests (Rose, 2007; Lappé & Landecker, 2015).
The ultimate consequence is a public health landscape shaped less by genuine health needs and more by the financial interests and strategic control of corporate stakeholders.
16.4 The Culmination of Control: Eugenics, Profit, Biotech Evolution, and the Threat of Universal Fortification
The intricate interplay between biochemical eugenics, population control, pharmaceutical profit, and biotechnology-driven evolution forms a deeply troubling convergence. Through systematically ignoring genetic vulnerabilities such as MTHFR polymorphisms, regulatory policies have subtly enforced a selective biochemical burden, disproportionately impacted certain populations and effectively functioned as covert eugenics (Marks, 2017; Rose, 2007). Concurrently, pharmaceutical corporations exploit these vulnerabilities, cultivating dependency and reaping immense profits from the resulting health crises (Angell, 2005 Goldacre, 2012).
The rapid deployment of biotechnological interventions, such as MRNA-based cancer vaccines, further amplifies this cycle, embedding biochemical dependence at a generational scale and subtly steering human evolution through biotech-mediated pathways (Lappé & Landecker, 2015).
The potential for extension of mandatory fortification to gluten-free products would represents an alarming intensification of this dynamic. Individuals who rely on gluten-free diets often have pre-existing autoimmune, neurological, or genetic conditions, precisely those most vulnerable to the detrimental effects of unmetabolised folic acid accumulation and disrupted methylation cycles (Lucock et al., 2022; Selhub & Rosenberg, 2016).
This universal biochemical control leaves vulnerable populations with no safe alternatives, further marginalizing genetic diversity, and entrenching biochemical dependency as normative healthcare practice. Unless critically challenged, this scenario will perpetuate generational harm and consolidate unprecedented pharmaceutical and biotech control over the very definition and future trajectory of human health and identity.
16.5 Emerging Questions: Potential Biochemical Interactions Between MRNA Vaccines and Synthetic Folic Acid Exposure
Recent advances in MRNA-based vaccines introduce novel mechanisms of rapid cellular protein synthesis, notably stimulating robust inflammatory and immune responses (Sahin & Türeci, 2021). Simultaneously, chronic exposure to synthetic folic acid (especially unmetabolized folic acid – UMFA) has been independently linked to accelerated proliferation of cancerous and precancerous cells, due to its critical role in DNA synthesis, methylation processes, and cell division (Kim, 2008; Mason et al., 2007; Selhub and Rosenberg, 2016).
The concurrent biochemical environments created by these two interventions – ongoing synthetic folic acid exposure and MRNA vaccine-induced protein production – could potentially synergize, accelerating cancer growth, particularly in genetically vulnerable or precancerous populations. This risk arises from the cumulative effects on inflammatory pathways, altered immune surveillance, and increased cellular metabolic activity, all conditions conducive to rapid tumour proliferation (Lucock et al., 2022; Sahin & Türeci, 2021).
Although direct evidence specifically examining interactions between MRNA vaccination and synthetic folic acid exposure remains scarce, existing biochemical knowledge strongly supports the plausibility of this scenario. Given the broad global implementation of both MRNA vaccines and mandatory folic acid fortification, the need for targeted research into these interactions is urgent. Such research could elucidate critical risks, inform public health guidelines, and ensure that medical interventions intended to improve health outcomes do not inadvertently amplify existing vulnerabilities.
17. Conclusion-The End of Blind Trust
This report has traced a deeply unsettling pattern: science selectively interpreted, data strategically suppressed, and public health policies shaped not by truth – but by profit and power. Folic acid fortification, long hailed as a public health triumph, now stands exposed as a dangerously oversimplified intervention built on outdated assumptions, omissions and flawed science. The evidence is copious, clear and had to be trimmed excessively for this report; this speaks for itself – and it is deafening.
Global pharmaceutical corruption is not a relic of the past – it is an ongoing assault on public health, manifest in policies that prioritize profit over life and deception over transparency. The evidence is as damning as it is comprehensive. From the hidden dangers of synthetic folic acid to the calculated obfuscation of scientific data, the report reveals a system engineered to conceal its own failures.
This analysis exposes how selective data presentation, regulatory capture, and industry-driven narratives have repeatedly endangered lives, particularly those most vulnerable. The very measures meant to protect – fortification policies, rebranded pharmaceuticals, and delayed warnings – have instead become instruments of control, masking the true cost in human lives and health.
As we stand at this crossroads, the choice is more of inevitability than decision; we can continue to accept a system that prioritizes profit over people, or we can demand a transformation – a return to transparency, accountability, and integrity in public health. The cost of silence is measured in lives; the cost of inaction, in irreversible damage and the time for change is now.
Public health has treated the human body as an identical collective, ignoring decades of data that clearly demonstrate our genetic, metabolic, and neurological diversity. For individuals with MTHFR mutations or impaired methylation pathways, this “preventive measure” was never safe; it was always a risk. A one-size-fits-all policy was never science -it was policy-based evidence in disguise.
The tragedy is not just in the biochemical damage, but in the systemic betrayal; regulatory bodies should have known about UMFA accumulation and yet have taken no action. Studies have published revelations, only to be buried beneath the narrative.
Governments have continued with fortification despite rising rates of cancer, immune dysfunction, and neurological decline; and a media structure that, rather than ask questions, became a mouthpiece for silence.
What has been sold as prevention may, in fact, be exposure. What was framed as “evidence-based” was evidence-filtered. A foundational flaw in many pro-folic acid studies is the inappropriate use of correlation as causation. A common pattern involves attributing reductions in neural tube defects (NTDs) solely to the introduction of folic acid fortification – while failing to account for significant confounding variables. These include the quiet withdrawal of known teratogens like thalidomide, valproate and paramethadione, advances in prenatal screening, and a marked rise in the detection and termination of pregnancies with diagnosed NTDs. When terminations are quietly excluded from NTD prevalence statistics, the illusion of success is maintained – but the reality is far more complex. Such logic should never be tolerated in clinical pharmacology or experimental medicine, yet it underpins decades of policy in nutritional science. Millions have been exposed to synthetic folic acid without personalised assessment or informed consent, despite growing evidence of genetic incompatibilities and long-term epigenetic effects. The result is not precision medicine -it is blunt-force policy and its consequences are only beginning to surface.
The cost of this silence is no longer theoretical. It is being measured in birth defects, autoimmune conditions, neurologically damaged children, cognitive decline, and cancers. These aren’t abstract risks, these are lives. The one-size-fits-all approach to folic acid fortification ignores critical genetic variability in folate metabolism. Instead of protecting vulnerable populations, these policies may be actively harming them -especially autistic individuals, who are disproportionately affected.
The most dangerous flaw in our medical system is the division of knowledge – Scientists design drugs without understanding their systemic fallout. Pharmacists dispense them without questioning their origin. Doctors prescribe them based on outdated protocols and limited understanding, and regulators approve them on corporate data. No one is accountable – because no one holds the full picture. That fragmentation does not appear to be accidental.
Genetic science has evolved, but policy has not. Despite the widespread availability of genomic data, the implications have been ignored. We now know that synthetic folic acid behaves differently in different bodies – yet we treat the population as a homogenous mass. That is not medicine; that is biochemical negligence.
Beyond this systemic abuse, here is a call to action. This report is not merely an academic exercise; it is a declaration that silence and complacency are no longer options. It demands that policymakers, health professionals, and the public re-evaluate long-standing practices and hold the institutions responsible for their failures. Every delayed warning, every manipulated study, every regulatory blind spot is a debt owed in lives lost and futures compromised.
This report is open for critical review, particularly by those with cross-disciplinary understanding – individuals who have navigated the intersection of genetics, biochemistry, pharmacology, toxicology, and public health policy. Only through such integrative scrutiny can we begin to uncover the full impact of systemic failure and move toward a model of health rooted in biological integrity, transparency, and truth.
This report is more than a collection of data – it is a call for reckoning and for the reclaiming of human rights that are being systematically breached. In this reckoning, there is no room for blind trust. The evidence forces us to confront the truth, however brutal it may be; and it is only by facing that truth that we can begin to rebuild a system that truly values life over profit.
This report was not produced within the confines of academia, but beyond them, to:
Halt all forms of mandatory folic acid fortification.
Investigate the regulatory bodies that greenlit it.
Demand public inquiry into the long-term consequences.
Reform a broken system that allows convenience and control to triumph over truth.
If no action is taken, we are not just complicit in ignorance. We are participants in biochemical eugenics – a quiet war against the vulnerable disguised as public health.

18. Action Summary for Researchers, Policymakers, and Institutions
This document has presented a comprehensive and multi-disciplinary analysis of synthetic Folic Acid exposure, genetic vulnerability, systemic regulatory failure, and the accelerating intersection of pharmaceutical dependency, epigenetic disruption, and health inequality. The implications are not only medical but ethical, social, and generational. For those positioned within academic, clinical, or policy institutions, the following actions represent critical next steps toward transparency, accountability, and safeguarding biological autonomy.
1. Reevaluate Fortification Policies Using Stratified Risk Models:
- Encourage public health bodies to integrate genetic stratification – particularly MTHFR polymorphism prevalence – into assessments of fortification safety.
2. Prioritize Research into UMFA Thresholds and Long-Term Accumulation Effects
- Advocate for urgent, independent research into the establishment of safe blood UMFA thresholds, with consideration for genetic subgroups.
3. Support Open Access to Raw Study Data and Conflict of Interest Declarations
- Promote transparency in all research related to folic acid, vaccines, and cancer epidemiology. Encourage journals and institutions to publish funding sources, affiliations, and potential bias frameworks.
Engage in Interdisciplinary Dialogue Between Nutritional Genomics, Oncology, and Autism Research
- Break down siloed research approaches by facilitating collaboration between Geneticists, Neurodevelopmental Researchers, Oncologists, and Epigeneticists to reevaluate assumptions about ‘safety’ and ‘risk’.
5. Establish Safe Dietary Pathways for Biochemically Vulnerable Populations
- Urge governments and industry to ensure that non-fortified, non-synthetic dietary options remain available and clearly labeled for populations with metabolic vulnerabilities
6. Track and Audit Cancer Vaccine Rollouts for Transparency and Post-Deployment Risk
- Demand clear documentation of clinical endpoints, fertility tracking, and long-term monitoring protocols for MRNA cancer vaccine program
7. Create a Safe Space for Scientific Dissent and Ethical Discourse
Ensure that concerns raised by independent researchers are not silenced by institutional allegiance, and that academic spaces remain open to critique of dominant pharmaceutical models.
Note for Public Release: A simplified, publicly accessible version of this document and action summary has been prepared. It will offer direct, clear steps for affected families, patients, and public advocates, while preserving the core factual basis of this report.
About the Author
Alexandra is a Psychology student at the Open University. She is autistic, ADHD, hyperlexic, and lives with chronic illness. Prior to her psychology studies, she worked for over a decade as a Pharmacy Technician in various settings. Alex now focuses on activism through academic writing, particularly in the areas of neuropsychology, genetics, and health science -fields closely connected to both her lived experience and specialist interests. She is currently preparing her first book on this topic for publication, which explores and expands these themes in greater depth.
References and Source List
The following includes all works cited directly in the text, as well as additional academic and investigative materials that informed the research and analysis in this report. Where not cited-in-line, these sources were still instrumental in the development of key arguments, context, and insight.
Abbott Laboratories (2012) ‘Abbott to pay $1.6 billion over Depakote marketing’, The Washington Post, 7 May. Available at: https://www.washingtonpost.com/business/economy/abbott-to-pay-16-billion-over-depakote-marketing/2012/05/07/gIQAp3F98T_story.html (Accessed: 2 April 2025).
Adab, N., Kini, U., Vinten, J., Ayres, J., Baker, G., & Clayton-Smith, J. (2004). The longer-term outcome of children born to mothers with epilepsy. Journal of Neurology, Neurosurgery & Psychiatry, 75(11), 1575-1583. `
Ahnen, D. J., Wade, S. W., Jones, W. F., Sifri, R., Mendoza Silveiras, J., Greenamyer, J., Guiffre, S., Axilbund, J. E., Spiegel, A. and You, Y. N., 2014. The increasing incidence of young-onset colorectal cancer: a call to action. Mayo Clinic Proceedings, 89(2), pp.216–224.
Al Aref, H. et al., 2024. Cerebral folate deficiency and metabolic epilepsies: diagnostic clues and early intervention outcomes. Annals of Pediatric Epileptology, 5(1), pp.1–12.
American Cancer Society (2024) Key statistics for prostate cancer. [online] Available at: https://www.cancer.org/cancer/types/prostate-cancer/about/key-statistics.html (Accessed 23 Mar. 2025).
American Cancer Society. (2024). Key Statistics for Colorectal Cancer. https://www.cancer.org/cancer/types/colon-rectal-cancer/about/key-statistics.html (Accessed 23 March, 2025)
American Cancer Society. (2024). Key Statistics for Colorectal Cancer. https://www.cancer.org/cancer/types/colon-rectal-cancer/about/key-statistics.html
American College of Obstetricians and Gynecologists (ACOG) (2017) ‘Practice Bulletin No. 187: Neural Tube Defects’, Obstetrics & Gynecology, 130(6), pp. e279–e290. Available at: https://www.acog.org/clinical/clinical-guidance/practice-bulletin/articles/2017/12/neural-tube-defects (Accessed: 27 March 2025).
American College of Obstetricians and Gynecologists (ACOG) (2017) ‘Practice Bulletin No. 187: Neural Tube Defects’, Obstetrics & Gynecology, 130(6), pp. e279–e290. Available at: https://www.acog.org/clinical/clinical-guidance/practice-bulletin/articles/2017/12/neural-tube-defects (Accessed: 27 March 2025).
American College of Obstetricians and Gynecologists (ACOG), 2017. Committee Opinion No. 700: Methods for estimating the due date. Obstetrics & Gynecology, 129(5), pp.e150–e154.
American Society for Reproductive Medicine (ASRM) (2016) Combined Hormonal Contraception and the Risk of Venous Thromboembolism: A Guideline. Available at: https://www.asrm.org/practice-guidance/practice-committee-documents/combined-hormonal-contraception-and-the-risk-of-venous-thromboembolism-a-guideline-2016 (Accessed: 4th April, 2025).
Angell M. (2005). The Truth About the Drug Companies: How They Deceive Us and What to Do About It. Random House.
Araghi, M., Soerjomataram, I., Bardot, A., Ferlay, J., Cabasag, C. J., Morrison, D. S., … & Bray, F. (2019). Changes in colorectal cancer incidence in seven high-income countries: a population-based study. The Lancet Gastroenterology & Hepatology, 4(7), 511-518.
Arnarson, A. (2024) ‘Folic acid vs folate – what’s the difference?’, Healthline, 15 April. Available at: https://www.healthline.com/nutrition/folic-acid-vs-folate (Accessed: 28 March 2025).
Arpino, C. et al. (2000) ‘Congenital malformations among infants born to women with epilepsy: a prospective study’, Reproductive Toxicology, 14(4), pp. 435–445.
Australian Institute of Health and Welfare, 2014. Neural tube defects in Australia: prevalence before mandatory folic acid fortification. Canberra: AIHW. Available at: https://www.aihw.gov.au/reports/mothers-babies/neural-tube-defects-prevalence-before-folic-acid/summary [Accessed 2 Apr. 2025].
Autism Speaks (2023) Medical Conditions Associated with Autism. Available at: https://www.autismspeaks.org/medical-conditions-associated-autism (Accessed 28th March, 2025)
Bailey, L.B. and Gregory, J.F. (1999) ‘Folate metabolism and requirements’, The Journal of Nutrition, 129(4), pp. 779–782.
Bailey, R.L., Dodd, K.W., Gahche, J.J., Dwyer, J.T., McDowell, M.A., Yetley, E.A., Sempos, C.A., Burt, V.L., Galuska, D.A., Ahuja, J.K.C. and Moshfegh, A.J., 2010. Total folate and folic acid intake from foods and dietary supplements in the United States: 2003–2006. The American Journal of Clinical Nutrition, 91(1), pp.231–237.
Bailey, S.W. and Ayling, J.E. (2009) ‘The extremely slow and variable activity of dihydrofolate reductase in human liver and its implications for high folic acid intake’, PNAS, 106(36), pp. 15424–15429.
Bailey, S.W. and Ayling, J.E. (2009) ‘The extremely slow and variable activity of dihydrofolate reductase in human liver and its implications for high folic acid intake’, Proceedings of the National Academy of Sciences, 106(36), pp. 15424–15429. Available at: https://doi.org/10.1073/pnas.0902072106 (Accessed: 1 April 2025).
Baker, G. A., et al. (2018) ‘Trends in antiepileptic drug use in pregnancy’, Epilepsia, 59(7), pp. 1191–1200.
Barker, J. and Galardi, T. (2011) ‘Autism, food allergy, and nutrition: Bringing the hidden voices of mothers into the mainstream debate’, Journal of Mother Studies, 3(2), pp. 31–48.
Bécares, N., et al. (2022) ‘Pregnancy outcomes following in utero exposure to valproate: A population-based cohort study’, British Journal of Clinical Pharmacology, 88(3), pp.1124–1135.
Bennett, L., Chen, Y. and Garcia, S., 2023. Environmental modulation of de novo mutation rates in neurodevelopmental disorders. Frontiers in Neuroscience, 17, p.112030. Available at: https://doi.org/10.3389/fnins.2023.112030 (Accessed 26 Mar. 2025).
Bennett, L., Chen, Y. and Garcia, S., 2023. Environmental modulation of de novo mutation rates in neurodevelopmental disorders. Frontiers in Neuroscience, 17, p.112030. Available at: https://doi.org/10.3389/fnins.2023.112030 (Accessed 26 Mar. 2025).
Berg, K. and Pineda, A. (2020) ‘Inequity by design: The social consequences of medically inaccessible foods’, Public Health Sociology, 18(3), pp. 145–162.
Berry, R. J., Li, Z., Erickson, J. D., Moore, C. A., Wang, H., Mulinare, J., Zhao, P., Wong, L. Y., & Gindler, J. (1999). Prevention of neural-tube defects with folic acid in China. The New England Journal of Medicine, 341(20), 1485-1490. https://doi.org/10.1056/NEJM199911113412001
Berry, R. J., Li, Z., Erickson, J. D., Moore, C. A., Wang, H., Mulinare, J., Zhao, P., Wong, L. Y., & Gindler, J. (1999). Erratum: Prevention of neural-tube defects with folic acid in China. The New England Journal of Medicine, 341(24), 1864. https://doi.org/10.1056/NEJM199912093412424
Best, K.E., Rankin, J. and Dolk, H. (2024) Neural tube defects in the context of folic acid fortification: current trends and unexplained plateaus. Journal of Public Health Nutrition, 27(1), pp. 34–44. doi:10.1017/S1368980024000212.
Biehl, J. and Petryna, A. (2013). When People Come First: Critical Studies in Global Health. Princeton University Press.
BioNTech. (2023). BioNTainer modular mRNA production facilities. Retrieved from https://en.wikipedia.org/wiki/BioNTech.
Blaylock, R.L., 2008. A possible central mechanism in autism spectrum disorders, part 1. Alternative Therapies in Health and Medicine, 14(6), pp.46–53.
Blusztajn, J.K. & Mellott, T.J., 2012. Choline nutrition programs brain development via DNA and histone methylation. Central Nervous System Agents in Medicinal Chemistry, 12(2), pp.82–94.
BMJ (2023) Ultra-Processed Foods and Cardiometabolic Health. Available at: https://www.bmj.com/content/383/bmj-2023-075294 (Accessed 23rd March, 2025).
BMJ Oncology (2023) Global burden of early-onset cancer from 1990 to 2019. Available at: https://bmjoncology.bmj.com/content/2/1/e000049 (accessed 23rd March, 2025).
Botto, L.D. and Yang, Q. (2000). 5,10-Methylenetetrahydrofolate reductase gene variants and congenital anomalies: a HuGE review. American Journal of Epidemiology, 151(9), pp.862–877.
Botto, L.D., Moore, C.A., Khoury, M.J. and Erickson, J.D. (2005) ‘Neural-tube defects’, New England Journal of Medicine, 341(20), pp. 1509–1519. doi:10.1056/NEJM199911113412006.
Bouboushian, J., 2012. Mom Blames Depakote for Son’s Birth Defect. Courthouse News Service, [online] 19 April. Available at: https://www.courthousenews.com/mom-blames-depakote-for-sons-birth-defect/ [Accessed 22 Mar. 2025].
Bower, C. and Stanley, F.J. (2004) ‘Epidemiology of neural tube defects and folic acid’, Fluids and Barriers of the CNS, 1(1), p.5. Available at: https://fluidsbarrierscns.biomedcentral.com/articles/10.1186/1743-8454-1-5 (Accessed: 27 March 2025).
Bower, C. and Stanley, F.J., 1989. Dietary folate as a risk factor for neural-tube defects: evidence from a case-control study in Western Australia. Medical Journal of Australia, 150(11), pp.613–619.
British Dietetic Association. (2023). Folic acid fortification in the UK. [online] Available at: https://www.bda.uk.com/resource/folic-acid-fortification-in-the-uk.html [Accessed 30 Mar. 2025].
British Medical Association and Royal Pharmaceutical Society, 1960s editions. British National Formulary (BNF). London: BMA and RPS. [Used for verification of absence of paramethadione in UK listings.]
Bromley, R.L., Mawer, G., & Clayton-Smith, J. et al. (2013). Cognitive outcomes following prenatal exposure to antiepileptic drugs: a prospective cohort study. The Lancet Neurology, 12(3), 244–252. https://doi.org/10.1016/S1474-4422(12)70323-X
Burnson, R. and Feeley, J. (2024) ‘Abbott loses $500 million infant formula verdict, lawyers say’, Bloomberg, 29 July. Available at: https://www.bloomberg.com/news/articles/2024-07-27/abbott-loses-500-million-infant-formula-verdict-lawyers-say (Accessed: 22 March 2025).
Burton, L.M. and Hardaway, C.R. (2012). Low-income mothers as ‘othermothers’: the social construction of parental fitness. Journal of Marriage and Family, 74(5), pp.1038–1053.
Business Insider. (2025). So many young people with colon cancer have clean diets. What gives? https://www.businessinsider.com/colon-cancer-environmental-causes-microplastics-antibiotics-sleep-2025-1
BusinessWire (2024). Global Gluten-Free Food Market Forecast to 2030 – Rising Incidence of Gluten Sensitivity and Increased Awareness Drive Growth. [online] Available at: https://www.businesswire.com/news/home/20240229005033/en/Global-Gluten-Free-Food-Market-Forecast-to-2030 (Accessed 28 Mar. 2025).
Calzolari, E., Bianchi, F., Rubini, M., Ritvanen, A. and Neville, A. (2004) ‘Epidemiology of cleft palate in Europe: implications for genetic research’, The Cleft Palate-Craniofacial Journal, 41(3), pp. 244–249. Available at: https://doi.org/10.1597/02-074 (Accessed: 2 April 2025).
Calzolari, E., Cocchi, G. and Neville, A.J. (2001) Report on periconceptional folic acid supplementation for Italy. European Commission Health & Consumer Protection Directorate-General. Available at: https://ec.europa.eu/health/ph_projects/2001/rare_diseases/fp_raredis_2001_a3_01_en.pdf (Accessed: 27 March 2025).
Canadian Medical Association Journal (CMAJ), 1959. Treatment of petit mal epilepsy in children: Comparison of trimethadione and paramethadione. Canadian Medical Association Journal, 81(4), pp.128–130.
Cancer Research UK (2023) Early-onset cancer: why are more young adults being diagnosed? Available at: https://news.cancerresearchuk.org/2023/01/24/early-onset-cancer-why-are-more-young-adults-being-diagnosed/ (Accessed 30th March, 20205)
Cancer Research UK (2024) Children’s cancer incidence statistics, Cancer Research UK. Available at: https://www.cancerresearchuk.org/health-professional/cancer-statistics/childrens-cancers/incidence
Cancer Research UK. (2020). Bowel cancer incidence statistics. [online] Available at: https://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/bowel-cancer/incidence [Accessed 30th March, 2025].
Cancer Research UK: Cancer Research UK (2023) Prostate cancer statistics. [online] Available at: https://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/prostate-cancer [Accessed 23 Mar. 2025].
Catassi, C. & Fasano, A., 2014. Celiac disease diagnosis: simple rules are better than complicated algorithms. American Journal of Medicine, 127(8), pp.691–693.
CDC (2023) Autism prevalence rises to 1 in 36 among U.S. children. Available at: https://www.cdc.gov/media/releases/2023/p0323-autism.html
CDC, 2015. Birth Defects COUNT: Data and Statistics on Neural Tube Defects. [online] Centers for Disease Control and Prevention. Available at: https://www.cdc.gov/ncbddd/birthdefects/ntd-data.html [Accessed 31 Mar. 2025].
CDC, 2023. Folic Acid: Facts and Recommendations. [online] Centers for Disease Control and Prevention. Available at: https://www.cdc.gov/folic-acid/index.html [Accessed 31 Mar. 2025].
CDC, 2023. MTHFR Gene Variant and Folic Acid Facts. [online] Centers for Disease Control and Prevention. Available at: https://www.cdc.gov/folic-acid/data-research/mthfr/index.html [Accessed 31 Mar. 2025].
CDC, 2023. Spina Bifida: Data and Statistics. [online] Centers for Disease Control and Prevention. Available at: https://www.cdc.gov/spina-bifida/data/index.html [Accessed 31 Mar. 2025].
Cedars-Sinai (2023) Autoimmune Diseases on the Rise: What to Know. Available at: https://www.cedars-sinai.org/newsroom/beverly-hills-courier-autoimmune-diseases-on-the-rise–what-to-know/
Centers for Disease Control and Prevention (2015) ‘Fortification of cereal-grain products with folic acid — United States, 1998–2011’, Morbidity and Mortality Weekly Report (MMWR), 64(1), pp. 1–5. Available at: https://www.cdc.gov/mmwr/preview/mmwrhtml/mm6401a2.htm (Accessed: 27 March 2025).
Centers for Disease Control and Prevention (2024) U.S. cancer statistics: Prostate cancer stat bite. [online] Available at: https://www.cdc.gov/united-states-cancer-statistics/publications/prostate-cancer-stat-bite.html [Accessed 23 Mar. 2025].
Centers for Disease Control and Prevention (2024) United States Cancer Statistics: Data Visualizations Tool. [online] Available at: https://www.cdc.gov/cancer/uscs/dataviz.html (Accessed 23 Mar. 2025).
Centers for Disease Control and Prevention (CDC) (1995) ‘Recommendations for the use of folic acid to reduce the number of cases of spina bifida and other neural tube defects’, Morbidity and Mortality Weekly Report, 44(RR-14), pp. 1–7. Available at: https://www.cdc.gov/mmwr/preview/mmwrhtml/00048410.htm (Accessed: 28 March 2025).
Centers for Disease Control and Prevention (CDC) (2023) Blood Clot Risk Factors: Pregnancy. Available at: https://www.cdc.gov/blood-clots/risk-factors/pregnancy.html (Accessed: 5th April, 2025).
Centers for Disease Control and Prevention (CDC), 1998. Recommendations for the use of folic acid to reduce the number of cases of spina bifida and other neural tube defects. Morbidity and Mortality Weekly Report (MMWR), 47(RR-3), pp.1–7. Available at: https://www.cdc.gov/mmwr/preview/mmwrhtml/00019479.htm
Centers for Disease Control and Prevention (CDC). (1992). Recommendations for the use of folic acid to reduce the number of cases of spina bifida and other neural tube defects.
Council of Europe, 1950. European Convention for the Protection of Human Rights and Fundamental Freedoms. Rome, 4 November 1950. Available at: https://www.echr.coe.int/documents/convention_eng.pdf [Accessed 6 Apr. 2025].
Centers for Disease Control and Prevention (CDC). (1998). Recommendations for the use of folic acid to reduce the number of cases of spina bifida and other neural tube defects. Morbidity and Mortality Weekly Report, 47(RR-8), 1-7.
Cermak, S.A., Curtin, C. and Bandini, L.G., 2010. Food selectivity and sensory sensitivity in children with autism spectrum disorders. Journal of the American Dietetic Association, 110(2), pp.238–246. https://doi.org/10.1016/j.jada.2009.10.032
Chapman, T. R. and Bilbo, S. D., 2019. The neuroimmune basis of MIA-linked neurodevelopmental disorders: From cells to circuits. Frontiers in Neuroscience, 13, p.1241.
Chen, A., Dietrich, K.N., Ware, J.H., Radcliffe, J. and Rogan, W.J., 2005. IQ and blood lead from 2 to 7 years of age: are the effects in older children the same as in young children? Environmental Health Perspectives, 113(5), pp.597–601.
Chen, J. and Reich, N.C., 2022. Cellular stress induces de novo synthetic lethality in cancer cells through modulation of interferon-stimulated genes. Cancer Research, 82(14), pp.2612–2623.
Chen, P.-C., Li, Y.-H., Lo, T.-H., Wu, Y.-C., Chang, W.-P., Chang, Y.-C. and Wang, I.-J. (2022) ‘Overview of Neural Tube Defects: Gene–Environment Interactions’, International Journal of Environmental Research and Public Health, 19(10), 6132. Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9138472/ (Accessed: 27 March 2025).
Choi, J.S., Han, J.Y., Park, Y.W., Ko, H.S., Yang, J.H. and Lee, Y. (2019) ‘Diagnostic accuracy of prenatal ultrasound for neural tube defects: a systematic review’, Obstetrics & Gynecology Science, 62(6), pp. 424–432. Available at: https://www.ogscience.org/upload/pdf/ogs-22263.pdf (Accessed: 27 March 2025).
Christensen, J. et al. (2013) ‘Prenatal valproate exposure and risk of autism spectrum disorders and childhood autism’, JAMA, 309(16), pp. 1696–1703. Available at: https://jamanetwork.com/journals/jama/fullarticle/1681408 (Accessed: 2 April 2025).
CIHR, NSERC & SSHRC, 2018. Tri-Council Policy Statement: Ethical Conduct for Research Involving Humans. [online] Government of Canada. Available at: https://ethics.gc.ca/eng/policy-politique_tcps2-eptc2_2018.html (Accessed 31 Mar. 2025).
Cochrane, N., Smith, D. and Jones, L., 2023. Functional folate deficiency: implications for health and disease. Journal of Nutritional Biochemistry, 108, p.109091.
Cochrane-backed endorsement of global fortification with limited genetic stratification.
Colapinto, C.K. et al. (2022) ‘High folic acid intake is associated with DNA instability in healthy adults’, Nutrients, 14(19), p. 3944. doi:10.3390/nu14193944.
Cole, B.F., Baron, J.A., Sandler, R.S., Haile, R.W., Ahnen, D.J., Bresalier, R.S., McKeown-Eyssen, G., Summers, R.W., Rothstein, R.I., Burke, C.A. and Snover, D.C., 2007. Folic acid for the prevention of colorectal adenomas: a randomized clinical trial. JAMA, 297(21), pp.2351–2359. https://doi.org/10.1001/jama.297.21.2351
Collet, T.H., Dubern, B., Mokhtari, M., et al. (2023) ‘Case report: Sibling pair with cerebral folate deficiency responsive to folinic acid therapy’, Molecular Genetics and Metabolism Reports, 36, 100941. Available at: https://www.sciencedirect.com/science/article/pii/S2214426923000216 (Accessed: 2 April 2025).
Colorectal Cancer Alliance. (2024). ACS Releases Colorectal Cancer Estimates for 2024. https://colorectalcancer.org/article/acs-releases-colorectal-cancer-estimates-2024
Comfort N. (2014). The Science of Human Perfection: How Genes Became the Heart of American Medicine. Yale University Press.
Cosey-Gay, F. et al. (2018). Child welfare system involvement among children with disabilities. Journal of Public Child Welfare, 12(1), pp.1–24.
Craciunescu, C.N., Brown, E.C., Mar, M.H., Albright, C.D. and Zeisel, S.H., 2004. Folic acid deficiency during late gestation decreases progenitor cell proliferation and increases apoptosis in fetal mouse brain. Journal of Nutrition, 134(1), pp.162–166.
Crider, K.S., Bailey, L.B. and Berry, R.J., 2011. ‘Folic acid food fortification – its history, effect, concerns, and future directions’. Nutrients, 3(3), pp.370–384. Available at: https://doi.org/10.3390/nu3030370 (Accessed 24th March, 2025).
Crider, K.S., et al. 2012. Unmetabolized folic acid, a potential marker of excessive intake.’ The American journal of Nutrition, 96(6), pp.1170-1177. Available at: https://doi.org/10.3945/ajcn.112.039511 (Accessed 23rd March, 2025).
Crider, K.S., Yang, T.P., Berry, R.J. and Bailey, L.B., 2012. Folate and DNA methylation: a review of molecular mechanisms and the evidence for folate’s role. Advances in Nutrition, 3(1), pp.21–38. Available at: https://doi.org/10.3945/an.111.000992 (Accessed 23rd March, 2025).
Critchfield, J.W., van Hemert, S., Ash, M., Mulder, L. and Ashwood, P., 2011. The potential role of probiotics in the management of childhood autism spectrum disorders. Gastroenterology Research and Practice, 2011, p.161358. https://doi.org/10.1155/2011/161358
Culty, M. (2023) Frontiers in Public Health Endocrine Disrupting Chemicals and Male Fertility. Frontiers in Public Health, 11. Doi:10.3389/fpubh.2023.1232646 Available at: https://www.frontiersin.org/articles/10.3389/fpubh.2023.1232646/full
Cumberlege, J. (2020) First Do No Harm: The report of the Independent Medicines and Medical Devices Safety Review. Available at: https://www.gov.uk/government/publications/independent-medicines-and-medical-devices-safety-review-report (Accessed: 2 April 2025).
D’Angelo, D., Loane, M., Nelen, V., Garne, E. and Dolk, H. (2017) ‘Trends in prevalence of neural tube defects in Europe from 1991 to 2011’, Birth Defects Research, 109(14), pp. 1030–1038. doi:10.1002/bdr2.1042.
D’Antoine, H., Bower, C., Stanley, F. and Elliott, E.J. (2019) ‘Folate status and neural tube defects in Aboriginal Australians: the success of mandatory fortification in reducing a health disparity’, Current Developments in Nutrition, 3. Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6642066/ (Accessed: 27 March 2025).
Dansky, L.V. and Finnell, R.H. (1991) ‘Valproic acid-induced neural tube defects in mice and the protective effect of folinic acid’, Teratology, 43(1), pp. 47–54.
De Rubeis, S., He, X., Goldberg, A.P., Poultney, C.S., Samocha, K. and Cicek, A.E., et al., 2014. Synaptic, transcriptional and chromatin genes disrupted in autism. Nature, 515(7526), pp.209–215. Available at: https://doi.org/10.1038/nature13772 [Accessed 26 Mar. 2025].
De Wals, P., Tairou, F., Van Allen, M.I., Uh, S.H., Lowry, R.B., Sibbald, B., Evans, J.A., Van den Hof, M.C., Zimmer, P., Crowley, M. and Fernandez, B., 2007. Reduction in neural-tube defects after folic acid fortification in Canada. New England Journal of Medicine, 357(2), pp.135-142.doi: 10.1056/NEJMoa067103.
Delcoigne, B. et al., 2020. Effects of wheat components on inflammatory markers: a narrative review. Nutrients, 12(5), pp.1356. https://doi.org/10.3390/nu12051356
Den Heijer, M., Koster, T., Blom, H.J., Bos, G.M., Briet, E., Reitsma, P.H., Vandenbroucke, J.P. and Rosendaal, F.R., 1996. Hyperhomocysteinemia as a risk factor for deep-vein thrombosis. New England Journal of Medicine, 334(12), pp.759–762.
Department of Health and Social Care (2024) Birth defects prevented by fortifying flour with folic acid. GOV.UK. Published 14 November 2024. Available at: https://www.gov.uk/government/news/birth-defects-prevented-by-fortifying-flour-with-folic-acid (Accessed: 28 March 2025).
De-Regil, L.M., Peña-Rosas, J.P., Fernández-Gaxiola, A.C. and Rayco-Solon, P., 2015. Effects and safety of periconceptional folate supplementation for preventing birth defects. Cochrane Database of Systematic Reviews, (12). DOI: 10.1002/14651858.CD007950.pub3
Desai, A., Sequeira, J.M. and Quadros, E.V. (2016) ‘The metabolic basis for developmental disorders due to defective folate transport’, Biochimie, 126, pp. 31–42. Available at: https://doi.org/10.1016/j.biochi.2016.02.012 (Accessed: 2 April 2025).
Digitale, E., 2016. Less decline than expected in rate of brain, spine defects after folic acid fortification program. Stanford Medicine News Center. [online] 18 May. Available at: https://med.stanford.edu/news/all-news/2016/05/less-decline-than-expected-in-rate-of-brain-spine-defects.html (Accessed 30 Mar. 2025).
Dolk, H., Loane, M. and Garne, E. (2008) ‘The prevalence of congenital anomalies in Europe’, Advances in Experimental Medicine and Biology, 686, pp. 349–364. doi:10.1007/978-90-481-9485-8_20.
Ebbing, M., Bønaa, K.H., Nygård, O., Arnesen, E., Ueland, P.M., Nordrehaug, J.E., Rasmussen, K., Nilsen, D.W.T., Refsum, H., Njølstad, I. et al. (2009). Cancer incidence and mortality after treatment with folic acid and vitamin B12. JAMA, 302(1
EFSA Panel on Nutrition, Novel Foods and Food Allergens (NDA). Tolerable upper intake level for folic acid. EFSA Journal. 2023;21(6): e08353. doi: 10.2903/j.efsa.2023.8353.
EFSA, 2006. Tolerable Upper Intake Levels for Vitamins and Minerals. [online] European Food Safety Authority. Available at: https://www.efsa.europa.eu/en/efsajournal/pub/314 [Accessed 31 Mar. 2025].
Electronic Medicines Compendium (EMC), 2000. Depakote 500 mg gastro-resistant tablets. Available at: https://www.medicines.org.uk/emc/product/1000/smpc [Accessed 2 Apr. 2025].
EUROCAT Morris, J.K., Addor, M.C., Ballestri, A., Barisic, I., Dolk, H., Garne, E., Haeusler, M., Khoshnood, B., O’Mahony, M., Queisser-Wahrendorf, A. and Rankin, J., 2015. BMJ, 351, p.h5949.
European Medicines Agency (2014) Assessment report under Article 31 of Directive 2001/83/EC: Valproate and related substances. EMA. Available at: https://www.ema.europa.eu/en/documents/referral/valproate-article-31-referral-assessment-report_en.pdf (Accessed: 27 March 2025).
European Medicines Agency (2018) Assessment report: Procedure under Article 31 of Directive 2001/83/EC – Valproate and related substances. Available at: https://www.ema.europa.eu/en/documents/referral/valproate-related-substances-article-31-referral-assessment-report_en.pdf (Accessed: 27 March 2025).
European Medicines Agency (EMA). (2009). Valproate and birth defect risks: Review findings.
Evidence of population-wide accumulation without metabolic screening.
Exley, C., Mold, M., Shardlow, E. and Linhart, C., 2018. Aluminium in brain tissue in autism. Journal of Trace Elements in Medicine and Biology, 46, pp.76–82. https://doi.org/10.1016/j.jtemb.2017.11.012
Exley, C., 2017. Aluminium and autism. The Hippocratic Post, [online] Available at: https://www.hippocraticpost.com/infection-disease/aluminium-and-autism/ [Accessed 15 Apr 2025].
Fardet, A. and Rock, E., 2021. Ultra-processed foods and chronic disease: A review of the epidemiological evidence. Nutrition Research Reviews, 34(1), pp.1–20. https://doi.org/10.1017/S0954422420000225
Fasano, A. et al., 2015. Non-celiac gluten sensitivity: the new frontier of gluten-related disorders. Nutrients, 7(3), pp.1925–1937.
FDA Ghost Archived page (ND): Drugs@FDA: FDA-Approved Drugs (Accessed 05/04/2025)
FDA (2013) FDA Drug Safety Communication: Valproate products contraindicated for migraine prevention in pregnant women due to decreased IQ scores in exposed children. U.S. Food and Drug Administration. Available at: https://www.fda.gov/drugs/drug-safety-and-availability (Accessed: 27 March 2025).
FDA (2023) Drugs@FDA: Sodium Valproate (Depakote, Depakote ER). Available at: https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm (Accessed: 27 March 2025).
Feldman, R. (2023). How Big Pharma Rigged the Patent System. The American Prospect.
Financial Times. (2023). Biotech startup Cellares scales up cell therapy production with advanced automation. Retrieved from https://www.ft.com/content/626007c4-9127-4682-9dab-154b32434029.
Food and Drug Administration (FDA). (2007). Depakote label update: Teratogenic risks and neural tube defects.
Forner, J., Kleinschmidt, D., Bock, R. et al. (2023). Targeted knockout of a conserved plant mitochondrial gene by genome editing. Nature Plants, 9, 1818–1831. https://doi.org/10.1038/s41477-023-01572-9
Frontiers in Nutrition (2024) Ultra-Processed Food Consumption and Metabolic Disease Risk. Available at: https://www.frontiersin.org/articles/10.3389/fnut.2024.1306310/full
Frontiers in Public Health (2024) ‘Endocrine Disruptor Chemicals Exposure and Female Fertility Declining’. 12.doi:10.3389/fpubh.2024.1466967. Available at: https://www.frontiersin.org/articles/10.3389/fpubh.2024.1466967/full
Frontiers in Public Health (2024) Endocrine Disruptor Chemicals Exposure and Female Fertility Declining. Available at: https://www.frontiersin.org/articles/10.3389/fpubh.2024.1466967/full
Frosst, P., et al. (1995). A candidate genetic risk factor for vascular disease: a common mutation in methylenetetrahydrofolate reductase. Nature Genetics, 10(1), pp.111–113.
Frye, R.E., et al. (2013). Folate receptor autoantibodies in autism spectrum disorders: a systematic review. Molecular Psychiatry, 18(3), pp.369–381.
Frye, R.E., Slattery, J., Delhey, L., Furgerson, B., Strickland, T. and Tippett, M., 2013. Folinic acid improves verbal communication in children with autism and language impairment: a randomized double-blind placebo-controlled trial. Molecular Psychiatry, 23(2), pp.246–256.
García-Bailo, B., El-Sohemy, A., Haddad, P. S., Arora, P., Benzaoui, N. and Karmali, M., 2012. Vitamins D, C, and E in the prevention of type 2 diabetes mellitus: Modulation of inflammation and oxidative stress. Journal of Nutrigenetics and Nutrigenomics, 5(2), pp.62–84
German, J., Kowal, A. and Ehlers, K.H. (1970) ‘Trimethadione and human teratogenesis’, Teratology, 3(4), pp. 349–362. Available at: https://doi.org/10.1002/tera.1420030412 (Accessed: 2 April 2025).
Gluten Free Watchdog, 2021. Folic acid in gluten-free foods: an underrecognized issue for sensitive individuals. [online] Available at: https://www.glutenfreewatchdog.org [Accessed 31 Mar. 2025].
Gluten-Free Watchdog, 2021. Folic acid in gluten-free foods: What consumers need to know. [online] Gluten Free Watchdog. Available at: https://www.glutenfreewatchdog.org/news/folic-acid-in-gluten-free-foods-what-consumers-need-to-know/ [Accessed 30 Mar. 2025].
GMC (2024) Good Medical Practice. General Medical Council. Available at: https://www.gmc-uk.org/ethical-guidance (Accessed: 4th March, 2025).
Goldacre B. (2012). Bad Pharma: How Drug Companies Mislead Doctors and Harm Patients. Fourth Estate.
González-Torres, L., Ramírez-Sandoval, J.C. and Rojas, L. (2024) ‘Food additives and their implication in inflammatory bowel disease and metabolic syndrome’, Clinical Nutrition, 43(2), pp. 311–318. Available at: https://www.sciencedirect.com/science/article/pii/S2405457724015122 (Accessed: 2 April 2025).
Goossens, J. F., Thuru, X., & Bailly, C. (2021). Properties and reactivity of the folic acid and folate photoproduct 6-formylpterin. Free Radical Biology and Medicine, 171, 1-10.
Grand View Research (2024). Gluten-Free Products Market Size, Share & Trends Analysis Report. [online] Available at: https://www.grandviewresearch.com/industry-analysis/gluten-free-products-market [Accessed 28 Mar. 2025].
Grand View Research, 2023. Gluten-Free Products Market Size, Share & Trends Analysis Report by Product, by Distribution Channel, by region, and Segment Forecasts, 2023 – 2030. [online] Available at: https://www.grandviewresearch.com/industry-analysis/gluten-free-products-market [Accessed 30 Mar. 2025].
Green, T.J. et al. (2005) ‘Plasma folate and unmetabolized folic acid in breast-fed and formula-fed infants’, The American Journal of Clinical Nutrition, 82(1), pp. 167–172.
Green, T.J., Skeaff, C.M., Rockell, J.E., Venn, B.J., Lambert, A., Todd, J., Khor, G.L. and Loh, S.P., 2005. Plasma folate and unmetabolized folic acid in older adults and infants consuming folic acid–fortified foods. The American Journal of Clinical Nutrition, 81(4), pp.1189S–1194S.
Greenblatt, J.M., Keller, A. and Chen, C.J. (2021) ‘Persistent prevalence of neural tube defects despite long-term folic acid fortification: a call for review’, BMJ Public Health, 5(9), pp. e1234–e1241. doi:10.1136/bmjph-2021-1234.
Hardon, A. and Moyer, E. (2014). Anthropology of pharmaceuticals: Ecologies of drug use. Annual Review of Anthropology, 43, pp.107–121.
Harvard Gazette (2024) Cancer keeps coming for the young. Why? Available at: https://news.harvard.edu/gazette/story/2024/02/cancer-keeps-coming-for-the-young-why/
Hazra, A., Kraft, P., Selhub, J., Giovannucci, E.L., Thomas, G., Hoover, R.N., Chanock, S.J. and Hunter, D.J., 2009. Common variants of FUT2 are associated with plasma vitamin B12 levels. Nature Genetics, 41(8), pp.932–935.
Health Canada (2007) ‘Recall of Depakene 500 mg and ratio-VALPROIC 500 mg due to formulation issues. Government of Canada Advisory, 11 May. Available at: Recall of two Valproic acid drugs, Depakene 500 mg and Ratio-Valproic 500 mg – Canada.ca
Health Council of the Netherlands. Risks of folic acid fortification. The Hague: Health Council of the Netherlands, 2000; publication no. 2000/12.
Health Foundation (2023) Rising cancer incidence in younger adults: what is going on? Available at: https://www.health.org.uk/features-and-opinion/blogs/rising-cancer-incidence-in-younger-adults-what-is-going-on
Healy, D. (2012) Pharmageddon. University of California Press
Hibbs, A.R. et al. (2016) ‘Excess folic acid impairs natural killer cell–mediated immunity in aged mice’, Journal of Immunology Research, Article ID 7865169.
Hirst, J., Tomlinson, J., & Baldwin, D. S. (2018) ‘Brand-name prescribing of valproate and the implications for data interpretation in teratogenic risk studies’, Drugs & Therapy Perspectives, 34(1), pp. 14–18.
House of Commons Health and Social Care Committee (2020) First Do No Harm: The Cumberlege Review. London: HMSO.
House of Commons Health and Social Care Committee (2021) Government response to the First Do No Harm report. HC 689. Available at: https://publications.parliament.uk/pa/cm5803/cmselect/cmhealth/689/report.html (Accessed: [insert date]).
House of Commons Health and Social Care Committee, 2020. Available at: https://publications.parliament.uk/pa/cm5801/cmselect/cmhealth/131/13102.htm [Accessed 2 Apr. 2025].
HRA (2021) CAREFOL-HT Study Summary. Health Research Authority. Available at: https://www.hra.nhs.uk/planning-and-improving-research/application-summaries/research-summaries/carefol-ht/ (Accessed: 2nd April, 2025)
Huang, X. et al. (2022) ‘High-dose folic acid intake reduces natural killer cell cytotoxicity in healthy adults: A randomized trial’, The Journal of Nutritional Biochemistry, 100, 108902.
Huh, S.Y., & Gordon, C.M. (2020). “Gluten-free diets and health: The critical intersection of privilege and policy.” Pediatrics, 145(5), e20194065.
ICMJE, 2019. Recommendations for the Conduct, Reporting, Editing, and Publication of Scholarly Work in Medical Journals. [online] Available at: http://www.icmje.org/recommendations/ [Accessed 31 Mar. 2025].
Independent Medicines and Medical Devices Safety Review (IMMDSR) (2020) First Do No Harm: The Report of the Independent Medicines and Medical Devices Safety Review. Available at: https://www.immdsreview.org.uk/downloads/IMMDSReview_Web.pdf (Accessed: 2 April 2025).
Institute of Medicine (IOM), 1998. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. Washington, DC: National Academies Press.
Investors. (2023). Moderna and Merck partner to rapidly produce personalized cancer vaccines using AI. Retrieved from https://www.investors.com/news/technology/moderna-stock-larry-ellison-ai-mrna-medicines-cancer-vaccines/.
Iossifov, I., O’Roak, B.J., Sanders, S.J., Ronemus, M., Krumm, N. and Levy, D., et al., 2014. The contribution of de novo coding mutations to autism spectrum disorder. Nature, 515(7526), pp.216–221. Available at: https://doi.org/10.1038/nature13908 [Accessed 26 Mar. 2025].
Jablonka E, Lamb MJ. (2014). Evolution in Four Dimensions: Genetic, Epigenetic, Behavioral, and Symbolic Variation in the History of Life. MIT Press.
James, S.J., et al. (2004). Metabolic biomarkers of increased oxidative stress and impaired methylation capacity in children with autism. The American Journal of Clinical Nutrition, 80(6), pp.1611–1617.
Jentink, J., Loane, M. A., Dolk, H., Barisic, I., Garne, E., Morris, J. K., … & EUROCAT Antiepileptic Study Working Group. (2010). Valproic acid monotherapy in pregnancy and major congenital malformations. New England Journal of Medicine, 362(23), 2185–2193. https://doi.org/10.1056/NEJMoa0907190
Johns Hopkins Bloomberg School of Public Health. (2016). Too much folate in pregnant women increases risk for autism, study suggests. Retrieved from https://publichealth.jhu.edu/2016/too-much-folate-in-pregnant-women-increases-risk-for-autism-study-suggests
Kalmbach, R.D., Choumenkovitch, S.F., Troen, A.P., D’Agostino, R., Jacques, P.F. and Selhub, J., 2008. Circulating folic acid in plasma: relation to folic acid fortification. The American Journal of Clinical Nutrition, 88(3), pp.763–768.
Katsuki, H., 2021. Valproate as a histone deacetylase inhibitor: neurobiological implications and clinical potential. Journal of Neural Transmission, 128(8), pp.1137–1145. doi:10.1007/s00702-021-02353-7.
Khan, J. et al. (2022) ‘Breast milk immunology and the developing infant immune system’, Frontiers in Immunology, 13, 901661.
Khoshnood, B., Loane, M., de Walle, H. and Dolk, H. (2015) ‘Long term trends in prevalence of neural tube defects in Europe: population-based study’, BMJ, 351, h5949. doi:10.1136/bmj.h5949.
Kim Y.I., (2008). Role of folate in colon cancer development and progression. Journal of Nutrition, 133(11), 3731S–3739S. doi:10.1093/jn/133.11.3731S.
Kim, Y.I., (2004). Folate and cancer: a tale of Dr. Jekyll and Mr. Hyde? The American Journal of Clinical Nutrition, 80(5), pp.1123–1124.
Kishi, T., Yoshimura, R., Ikuta, T., Iwata, N. and Hashimoto, R., 2013. Brain-derived neurotrophic factor and major depressive disorder: Evidence from meta-analyses. Frontiers in Psychiatry, 4, p.147.
Kliegman, R.M. et al., 2016. Nutritional neurologic disorders in infants and children: clinical impact of vitamin B12 deficiency. Paediatrics in Review, 37(9), pp.364–370. (PMID: 27589994)
Kowalski, T.W., Senger, M.R., and Bogo, M.R. (2016) ‘A mechanistic view of the teratogenicity of thalidomide: implications for embryonic development and neurotoxicity’, Toxicology Letters, 258, pp.210–220.
Krishnaveni, G.V. and Srinivasan, K., 2021. Folate in brain development and disease. Indian Journal of Clinical Biochemistry, 36(2), pp.117–125.
Lammer, E.J. et al. (1991) ‘Maternal use of valproic acid and risk for spina bifida’, New England Journal of Medicine, 324(19), pp. 1335–1338.
Landrigan, P.J., 2010. What causes autism? Exploring the environmental contribution. Current Opinion in Paediatrics, 22(2), pp.219–225.
Lappé M, Landecker H. (2015). Sociology in an age of genomic instability: Copy number variation, somatic mosaicism, and the fallen genome. Advances in Medical Sociology, 16, 157–186. doi:10.1108/S1057-629020150000016008
Lee, A.R., Ng, D.L., Zivin, J., & Green, P.H.R. (2007). “Economic burden of a gluten-free diet.” Journal of Human Nutrition and Dietetics, 20(5), 423–430.
Lee, H.J., Popkin, B.M. and Kim, S. (2007) ‘The burden of synthetic nutrient fortification in processed wheat foods’, Public Health Nutrition, 10(6), pp. 560–568.
Leitner, Y. (2014). The co-occurrence of autism and attention deficit hyperactivity disorder in children – what do we know? Frontiers in Human Neuroscience, 8, 268.
Lexchin, J. (2005). Drug withdrawals from the Canadian market for safety reasons, 1963–2004.
Lexchin, J. (2015) ‘How safe are new drugs? Market withdrawal of drugs approved in Canada between 1990 and 2009’, Open Medicine, 9(1), pp. 24–29.
Lexchin, J. (2020). Evergreening and the Pharmaceutical Industry. Journal of Law, Medicine & Ethics, PMC7592140.
Li, Y., et al. (2020). Association between MTHFR C677T polymorphism and autism spectrum disorder: A meta-analysis. BMC Pediatrics, 20(1), 1–9. https://bmcpediatr.biomedcentral.com/articles/10.1186/s12887-020-02330-3
Lietman, P.S., Zackai, E.H., Mellman, W.J., Neiderer, B. and Hanson, J.W. (1975) ‘The fetal trimethadione syndrome’, The Journal of Pediatrics, 87(2), pp. 280–284. Available at: https://doi.org/10.1016/S0022-3476(75)80603-2 (Accessed: 2 April 2025).
Light, D.W., Lexchin, J. & Darrow, J.J. (2013). Institutional Corruption of Pharmaceuticals and the Myth of Safe and Effective Drugs. Journal of Law, Medicine & Ethics, 41(3), 590–600. https://doi.org/10.1111/jlme.12068
Lucock M, Yates Z, Veysey M, et al. A critical examination of the role of dietary folate in health. Critical Review Food Science Nutrition. 2022;62(10):2572–2587. doi:10.1080/10408398.2020.1857333.
Lucock M. (2000). Folic acid: nutritional biochemistry, molecular biology, and role in disease processes. British Journal of Biomedical Science, 57(2): 84–93.
Lucock, M. et al., 2013. Folic acid–vitamin therapy and genetic polymorphisms: evidence for a significant gene–nutrient interaction in relation to neural tube defects. Molecular Genetics and Metabolism, 110(3), pp.303–310.
Lucock, M., Yates, Z., Veysey, M., Beckett, E. and Martin, C., 2014. Folic acid fortification: a double-edged sword. Current Opinion in Clinical Nutrition & Metabolic Care, 17(4), pp.288–293.
Lundin, K.E.A. & Wijmenga, C., 2015. Coeliac disease and autoimmune disease—genetic overlap and screening. Nature Reviews Gastroenterology & Hepatology, 12(9), pp.507–515.
Maenner, M.J., Shaw, K.A., Bakian, A.V., Bilder, D.A., Durkin, M.S. and Esler, A., et al., 2023.
Mai, C.T., Isenburg, J.L., Appaiahgari, M.B., Farr, S.L., Ethen, M.K., Rickard, R., Gilboa, S.M. and Honein, M.A., 2024. National population-based estimates for major birth defects, United States, 2016–2020. Atlanta: Centers for Disease Control and Prevention. Available at: https://stacks.cdc.gov/view/cdc/149716 (Accessed 31 Mar. 2025).
Mai, C.T., Williams, J., Mulinare, J., Isenburg, J., Flood, T.J., Ethen, M., Frohnert, B. and Kirby, R.S., 2015. Updated estimates of neural tube defects prevented by mandatory folic acid fortification—United States, 1995–2011. Morbidity and Mortality Weekly Report (MMWR), 64(1), pp.1–5. Available at: https://www.cdc.gov/mmwr/preview/mmwrhtml/mm6401a2.htm (Accessed 2 Apr. 2025).
Market Research Future (2024) Gluten-Free Products Market Size Worth $13.67 Billion by 2030. Available at: https://www.marketresearchfuture.com/reports/gluten-free-products-market-1605
Marks J. (2017). Is Science Racist? Debating Race. Polity Press.
Martin, M. (2017) Dépakine, le Scandale: je ne pouvais pas me taire. Paris: Éditions L’Harmattan.
Mason, J.B., Dickstein, A., Jacques, P.F., Haggarty, P., Selhub, J., Dallal, G. and Rosenberg, I.H., 2007. A temporal association between folic acid fortification and an increase in colorectal cancer rates may be illuminating important biological principles: a hypothesis. Cancer Epidemiology, Biomarkers & Prevention, 16(7), pp.1325–1329. https://doi.org/10.1158/1055-9965.EPI-07-0329
Matzke, M.A. and Mosher, R.A., 2014. RNA-directed DNA methylation: an epigenetic pathway of increasing complexity. Nature Reviews Genetics, 15(6), pp.394–408.
Mayo Clinic (2023) Celiac Disease Prevalence Remains High. Available at: https://newsnetwork.mayoclinic.org/discussion/new-study-calculates-autoimmune-disease-prevalence-in-u-s/
Mayo Clinic News Network (2023) New study calculates autoimmune disease prevalence in U.S. Available at: https://newsnetwork.mayoclinic.org/discussion/new-study-calculates-autoimmune-disease-prevalence-in-u-s/
McBride, W.G., 1961. Thalidomide and congenital abnormalities. The Lancet, 278(7216), p.1358.
McDonnell, R., Johnson, H. and Delany, V. (1991) ‘The changing incidence of neural tube defects in Scotland’, BMJ. Available at: https://pubmed.ncbi.nlm.nih.gov/2061799/ (Accessed: 27 March 2025).
McDonnell, R., Johnson, Z., Delany, V., and O’Reilly, E. (1999) ‘Neural tube defects in the Republic of Ireland in 1996–1997’, Irish Medical Journal, 92(3), pp.100–102.
McKay, J.A. et al., 2002. Folate depletion during pregnancy and lactation reduces genomic DNA methylation in murine offspring. Proceedings of the National Academy of Sciences, 99(8), pp.5606–5610.
McNulty, H., Pentieva, K., Hoey, L., Ward, M., McNulty, B.A. and Strain, J.J., 2013. Emerging issues in folate and related B vitamin research: update on folate and neural tube defect prevention. Nutrition Research Reviews, 13(2), pp.215–244. Available at: https://pubmed.ncbi.nlm.nih.gov/23719554/ (Accessed 23rd March, 2025)
MDPI Nutrients (2024) Association Between Ultra-Processed Food Consumption and Non-Alcoholic Fatty Liver Disease. Available at: https://www.mdpi.com/2072-6643/16/20/3524
Meador, K.J. et al. (2006) ‘Cognitive function at 3 years of age after foetal exposure to antiepileptic drugs’, New England Journal of Medicine, 360(16), pp. 1597–1605.
Meador, K.J. et al. (2013) ‘Fetal antiepileptic drug exposure and cognitive outcomes at age 6 years (NEAD study)’, Lancet Neurology, 12(3), pp. 244–252.
Meador, K.J., Baker, G.A., Browning, N. et al. (2009). Cognitive function at 3 years of age after foetal exposure to antiepileptic drugs. The New England Journal of Medicine, 360(16), 1597–1605. https://doi.org/10.1056/NEJMoa0803531
Meador, K.J., Pennell, P.B., Harden, C.L., Gordon, J.C., Tomson, T. and Kaplan, P.W. (2006) ‘Pregnancy registries in epilepsy: a consensus statement on health outcomes’, Neurology, 67(3), pp. 408–412.
Medicines and Healthcare products Regulatory Agency (MHRA) (2021) Valproate use by women and girls: new regulatory measures. Available at: https://www.gov.uk/drug-safety-update/valproate-use-by-women-and-girls-new-regulatory-measures (Accessed: 27 March 2025).
Medicines and Healthcare products Regulatory Agency (MHRA) (2023) ‘Valproate use by women and girls: new regulatory measures’. Available at: https://www.gov.uk/drug-safety-update/valproate-use-by-women-and-girls-new-regulatory-measures (Accessed: 27 March 2025).
Mesalles-Naranjo, O., Grant, I., Wyper, G.M.A., Stockton, D., Dobbie, R. and McFadden, M. (2018) ‘Trends and inequalities in the burden of mortality in Scotland 2000–2015’, PLOS ONE, 13(8), e0196906. Available at: https://doi.org/10.1371/journal.pone.0196906 (Accessed: 2 April 2025).
MHRA (2009) Sodium valproate: updated guidance on use in pregnancy. Medicines and Healthcare products Regulatory Agency. Available at: https://www.gov.uk/drug-safety-update/valproate-updated-guidance (Accessed: 27 March 2025).
MHRA (2015) Valproate and the risk of abnormal pregnancy outcomes: letter to healthcare professionals. Medicines and Healthcare products Regulatory Agency. Available at: https://www.cas.mhra.gov.uk/ViewandAcknowledgment/ViewAttachment.aspx?Attachment_id=102094 (Accessed: 3rd April, 2025).
MHRA (2015) Valproate and the risk of abnormal pregnancy outcomes: letter to healthcare professionals. Medicines and Healthcare products Regulatory Agency. Available at: https://www.cas.mhra.gov.uk/ViewandAcknowledgment/ViewAttachment.aspx?Attachment_id=102094 (Accessed: 3rd April, 2025).
Milton, D., Heasman, B. & Gillespie-Lynch, K., 2019. The double empathy problem: Ten years on. Frontiers in Psychology, 10, p.2676. Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6512057/
Moderna. (2023). Moderna’s mRNA pipeline. Retrieved from https://www.modernatx.com/research/pipeline.
Modgil, R., Gopinathan, P. and Sharma, M. (2022) ‘Data availability and transparency in global folic acid policy evaluation’, International Journal of Epidemiology, 51(5), pp. 1278–1287. doi:10.1093/ije/dyac115.
Moll, S. and Varga, E.A. (2015) ‘Homocysteine and MTHFR Mutations’, Circulation, 132(1), pp. e6–e9.
Molloy, A.M. (2008) ‘Should folic acid be fortified into all wheat flour?’, The American Journal of Clinical Nutrition, 88(2), pp. 253–254. Available at: https://pubmed.ncbi.nlm.nih.gov/18615588/ (Accessed: 2 April 2025).
Molloy, A.M., Kirke, P.N. and Scott, J.M. (2023) ‘Time to reassess folic acid’s population impact: are we measuring the right outcomes?’, Journal of Medical Ethics and Health Policy, 19(4), pp. 210–218. doi:10.1016/j.jmehp.2023.04.002.
Monteiro, C.A., Cannon, G., Levy, R.B., Moubarac, J.C., Jaime, P., Martins, A.P., Canella, D., Louzada, M. and Parra, D., 2019. Ultra-processed foods: What they are and how to identify them. Public Health Nutrition, 22(5), pp.936–941. https://doi.org/10.1017/S1368980018003762
Montgomery v Lanarkshire Health Board [2015] UKSC 11. Available at: https://www.supremecourt.uk/cases/docs/uksc-2013-0136-judgment.pdf (Accessed: [insert date]).
Moore, S. J., Turnpenny, P., Quinn, A., Glover, S., Lloyd, D. J., Montgomery, T., & Dean, J. C. (2000). A clinical study of 57 individuals with fetal anticonvulsant syndrome. Journal of Medical Genetics, 37(7), 489-497.
Moretti, P., Peters, S.U., Del Gaudio, D., Sahoo, T. & Zori, R.T., 2005. Cerebral folate deficiency with leukoencephalopathy and microangiopathy: improvement with folinic acid therapy. Archives of Neurology, 62(5), pp.666–670.
Morris, J.K., Addor, M.C., Ballestri, A., Barisic, I., Dolk, H., Garne, E., Haeusler, M., Khoshnood, B., O’Mahony, M., Queisser-Wahrendorf, A. and Rankin, J.,EUROCAT 2015. BMJ, 351, p.h5949.
McNulty, B., McNulty, H., Marshall, B., Ward, M., Molloy, A.M., Scott, J.M., et al. (2013). Impact of continuing folic acid after the first trimester of pregnancy: findings of a randomized trial of Folic Acid Supplementation in the Second and Third Trimesters. The American Journal of Clinical Nutrition, 98(1), 92–98. https://doi.org/10.3945/ajcn.112.057489
MMWR Morbidity and Mortality Weekly Report, 41(RR-14), 1–7. https://www.cdc.gov/mmwr/preview/mmwrhtml/00019479.htm
MRC Vitamin Study Research Group. (1991). Prevention of neural tube defects: results of the Medical Research Council Vitamin Study. Lancet, 338(8760), 131–137. https://doi.org/10.1016/0140-6736(91)90133-A
Nakamura, A., Suzuki, Y., Tanaka, M., and Abe, K., 2020. Valproate exposure during early pregnancy: A critical period when folate is essential for neural tube closure. Congenital Anomalies, 60(6), pp.169–176. doi:10.1111/cga.12345.
Nanishi, K., Green, J., Taguri, M., Jimba, M. and Kiriya, J. (2019) ‘Prevalence of neural tube defects and folic acid supplementation in Japan: Results from a multicenter birth cohort’, Birth Defects Research, 111(7), pp. 500–507. Available at: https://pubmed.ncbi.nlm.nih.gov/30883906/ (Accessed: 27 March 2025)
Naseh, M. et al. (2024) ‘Folate-to-vitamin B12 ratio and adverse pregnancy outcomes: A prospective cohort study’, Nutrients, 16(18), p. 3194. doi:10.3390/nu16183194.
National Cancer Institute (2024) Cancer stat facts: Prostate cancer. Surveillance, Epidemiology, and End Results Program (SEER). [online] Available at: https://seer.cancer.gov/statfacts/html/prost.html [Accessed 23 Mar. 2025].
National Health Council (2023) A Major Health Crisis: The Alarming Rise of Autoimmune Disease. Available at: https://nationalhealthcouncil.org/blog/a-major-health-crisis-the-alarming-rise-of-autoimmune-disease/
National Health Council (2023) A Major Health Crisis: The Alarming Rise of Autoimmune Disease. Available at: https://nationalhealthcouncil.org/blog/a-major-health-crisis-the-alarming-rise-of-autoimmune-disease/
National Health Service (NHS), UK (2022) Vitamins and minerals – Folate. Available at: https://www.nhs.uk/conditions/vitamins-and-minerals/folic-acid/ (Accessed: 28 March 2025).
National Institute for Health and Care Excellence (NICE), 2015. Coeliac disease: recognition, assessment and management (NG20). London: NICE.
NCATS. Paramethadione Compound Profile. NCATS Z615FRW64N
Nettleton, S. (2020) ‘Food, class and stigma in health discourse: From wellness to pseudoscience’, Journal of Critical Public Health, 30(1), pp. 59–74.
New York Post (2024) Autism diagnoses are skyrocketing in the US, new study finds. Available at: https://nypost.com/2024/10/31/lifestyle/autism-diagnoses-are-skyrocketing-in-the-us-new-study/
New York Post. (2025). Why colorectal cancer is often only caught in late stages – as rates under 45 are on the rise. https://nypost.com/2025/03/07/health/why-colorectal-cancer-is-often-only-caught-in-late-stages/
NHS (2023) Vitamins, supplements and nutrition in pregnancy. Available at: https://www.nhs.uk/pregnancy/keeping-well/vitamins-supplements-and-nutrition/ (Accessed: [insert date]).
NHS, (2024). Folic acid. [online] NHS.uk. Available at: https://www.nhs.uk/conditions/vitamins-and-minerals/folic-acid/ [Accessed 31 Mar. 2025].
O’Connor, D.L., Andrews, W.L., Hayward, S., Picciano, M.F., Dunn, R.L., Hartmann, E.E. and Hartmann, P.E., 2013. Nutrient intakes and growth of exclusively breast-fed infants. The Journal of Pediatrics, 143(4), pp.686–690.
Oakley, G.P. (1997). Folate fortification: time for action. BMJ, 315(7105), pp.769–770.
Oakley, G.P. (1999). Folic acid-preventable spina bifida and anencephaly. Birth Defects Research, 53(7), pp.588–592.
Oakley, G.P. (2002). Delaying folic acid fortification puts women and children at risk of preventable harm. BMJ, 324(7345), pp.1348–1349.
Obeid, R. and Herrmann, W. (2012). The role of folic acid and vitamin B12 in the pathogenesis and progression of neurodegenerative diseases. Current Pharmaceutical Design, 19(4), pp. 486–498.
Obeid, R. et al., 2015. Unmetabolized folic acid: relation to folic acid fortification and risks. Clinical Chemistry and Laboratory Medicine, 53(5), pp.679–687.
Obeid, R., & Herrmann, W. (2019). Unmetabolized folic acid: Relation to autism, cancer, and masking of B12 deficiency. Nutrients, 11(3), 593. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6537060/
O’Connor, D.L. et al. (2013) ‘Prevalence and predictors of high folate and cobalamin status in Canadian infants’, The Journal of Nutrition, 143(2), pp. 196–202.
OECD (2024) Declining Fertility Rates Put Prosperity of Future Generations at Risk. Available at: https://www.oecd.org/newsroom/declining-fertility-rates-put-prosperity-of-future-generations-at-risk.htm
Oklahoma State University Extension (2023). Going Gluten-Free: Fad, Fact, or Health Necessity? [online] Available at: https://extension.okstate.edu/fact-sheets/going-gluten-free-fad-fact-or-health-necessity.html (Accessed 28 Mar. 2025).
OMIM (2024). MTHFR – Methylenetetrahydrofolate Reductase. Online Mendelian Inheritance in Man, Entry #607093. Available at: https://omim.org/entry/607093 (Accessed: (4th April, 2025).
Ondičová, M., Irwin, R.E., Thursby, S.-J., Hilman, L., Caffrey, A., Cassidy, T., McLaughlin, M., Lees-Murdock, D.J., Ward, M., Murphy, M., Lamers, Y., Pentieva, K., McNulty, H. and Walsh, C.P. (2022) ‘Folic acid intervention during pregnancy alters DNA methylation, affecting neural target genes through two distinct mechanisms’, Clinical Epigenetics, 14(1), p.63. Available at: https://doi.org/10.1186/s13148-022-01282-y (Accessed: 1 April 2025).
OpenSecrets. (2024) AbbVie Inc: Lobbying profile and expenditures. Center for Responsive Politics. Available at: https://www.opensecrets.org/orgs/abbvie-inc/lobbying?id=D000066804 (Accessed: 2 April 2025).
Paramethadione – Uses, DMF, Dossier, Manufacturer, Supplier, Licensing, Distributer, Prices, News, GMP Available at: Paramethadione: Uses, Dosage, Side Effects, Food Interaction & FAQ
Pearson, H., 2005. Pervasive vitamin fortification could alter genes. Nature, [online] Available at: https://www.nature.com/articles/news050328-10
Pfeiffer, C.M., Sternberg, M.R., Fazili, Z., Yetley, E.A., Lacher, D.A., Bailey, R.L., Johnson, C.L. (2015). Unmetabolized folic acid is detected in nearly all serum samples from US children, adolescents, and adults. The Journal of Nutrition, 145(3), pp.520–531.
PHIRDA (Pharmaceutical Innovation and Research Development Association of China). (2021). Overview of SFDA Drug Approvals. [in Chinese] Retrieved from https://www.phirda.com/artilce_18813.html
Physicians’ Desk Reference (PDR), 1954. Paradione (paramethadione). 8th ed. Montvale, NJ: Medical Economics Company, pp.1042–1043.
Plumptre, L., Masih, S.P., Ly, A., Aufreiter, S., Sohn, K.J., Croxford, R., Lausman, A.Y., Berger, H., O’Connor, D.L. and Kim, Y.I. (2015). High concentrations of folate and unmetabolized folic acid in a cohort of pregnant Canadian women and umbilical cord blood. The American Journal of Clinical Nutrition, 102(4), pp.848–857.
Pohl, H., Tzianetas, R. and Reilly, J. (2021) ‘Cost barriers to gluten-free diets in vulnerable populations’, Journal of Food Policy, 54(4), pp. 88–104.
Prentice, A. et al. (2002) ‘Thymus size and breast-feeding: Evidence for the influence of early diet on the immune system’, Acta Paediatric, 91(1), pp. 54–56.
Prevalence and disparities in the detection of autism without intellectual disability. Pediatrics, 151(2), e2022056594. Available at: https://doi.org/10.1542/peds.2022-056594 [Accessed 26 Mar. 2025].
Prostate Cancer and Prostatic Diseases, 17(3), pp.213–219. https://doi.org/10.1038/pcan.2014.17
Pu, D., et al. (2023). Genetic and metabolic markers in autism: MTHFR mutation and folate metabolism. Genes, 14(3), 709. https://www.mdpi.com/2073-4425/14/3/709
Puga, A.M., Ruperto, M., Samaniego-Vaesken, M. de L., Montero-Bravo, A., Partearroyo, T. and Varela-Moreiras, G., 2021. Effects of supplementation with folic acid and its combinations with other nutrients on cognitive impairment and Alzheimer’s disease: A narrative review. Nutrients, 13(9), p.2966. Available at: https://doi.org/10.3390/nu13092966 [Accessed 31 Mar. 2025].
Qian 2023 Uncovering the hidden dangers and molecular mechanisms of excessive folic acid intake’, Frontiers in Nutrition, 10, 1064840. https://doi.org/10.3389/fnut.2023.1064840
Quadros, E.V., Sequeira, J.M. and Brown, R.T., 2016. Folate receptor autoantibodies and autoimmunity: significance in health and disease. Biochimie, 126, pp.52–61. Available at: https://doi.org/10.1016/j.biochi.2016.08.008 [Accessed 31 Mar. 2025].
Québec E&M Working Group. (2019). Management of epilepsy in pregnancy. GEMOQ Guidelines. https://gemoq.ca/wp-content/uploads/2021/12/management-of-epilepsy-in_pregnancy2019.pdf
Quinlivan, E.P. and Gregory, J.F. (2008). DNA integrity and methylation. In: Bailey, L.B. (Ed.), Folate in Health and Disease, 2nd ed. CRC Press, pp.295–320.
Raghavan, R., Selhub, J., Paul, L., Ji, Y., Wang, G., Hong, X., Pearson, C., Ortiz, M., Burd, I., Goldenberg, C., Wang, X. (2020). A prospective birth cohort study on cord blood folate subtypes and risk of autism spectrum disorder. American Journal of Clinical Nutrition, 112(5), 1304–1317. https://doi.org/10.1093/ajcn/nqaa208
Rai, V., 2020. Association of MTHFR C677T polymorphism with autism: Evidence from a meta-analysis. The Egyptian Journal of Medical Human Genetics, 21(1), pp.1–7.
Ramaekers, V.T. and Rothenberg, S.P., 2013. Autoantibodies against folate receptors in autism spectrum disorder. Drug Discovery Today: Disease Models, 10(1), pp.e43–e47.
Ramaekers, V.T., Blau, N., Sequeira, J.M. and Quadros, E.V., 2007. Cerebral folate deficiency and CNS disturbances. Developmental Medicine & Child Neurology, 49(10), pp.803–805.
Ramaekers, V.T., Blau, N., Sequeira, J.M., Nassogne, M.C. and Quadros, E.V., 2005. Folate receptor autoimmunity and cerebral folate deficiency in low-functioning autism with neurological deficits. Neuropediatrics, 36(06), pp.326–331.
Ramaekers, V.T., Rothenberg, S.P., Sequeira, J.M., Opladen, T., Blau, N. and Quadros, E.V., 2007. Autoantibodies to folate receptors in the cerebral folate deficiency syndrome. New England Journal of Medicine, 357(12), pp.1245–1252.
Ray, J.G. et al. (2002). Folic acid food fortification in Canada: update on health outcomes. Canadian Medical Association Journal, 167(5), pp.465–472.
Refsum H, Smith AD, Ueland PM, et al. (2022). Facts and recommendations about total homocysteine determinations: an expert opinion. Clinical Chemistry, 68(5), 648–660. doi:10.1093/clinchem/hvac022
Research and Markets, 2023. Global Gluten-Free Food Market Report 2023-2032. [online] Available at: https://www.researchandmarkets.com [Accessed 31 Mar. 2025].
Reuters (2024) Global Fertility Rates Decline, Shifting Population Burden to Low-Income Countries. Available at: https://www.reuters.com/world/global-fertility-rates-decline-shifting-population-burden-low-income-countries-2024-03-20/
Reynolds, E.H., Bailey, S.J. and Bottiglieri, T., 2022. Antiepileptic drugs, folate one-carbon metabolism, genetics, and epigenetics: Congenital, developmental, and neuropsychological risks and antiepileptic action. Epilepsia, 63(12), pp.3089–3104. doi:10.1111/epi.17406.
Riley, K.D., Hastings, J.W., Hennessy, B.A., et al. (2021). Hispanic women’s perceptions of neural tube defects and folic acid: Implications for fortification in the U.S. Journal of Nutrition Education and Behavior, 53(5), pp.395–403.
Risks of folic acid fortification. The Hague: Health Council of the Netherlands. Available at: https://www.healthcouncil.nl/documents/advisory-reports/2000/11/15/risks-of-folic-acid-fortification (Accessed: 1 April 2025).
Robert, E., Guibaud, P., & Morichon-Delvallez, N. (1983). Valproic acid and spina bifida. Lancet, 2(8355), 1392.
Roffman, J.L. et al., 2011. Genetic variation throughout the folate metabolic pathway and its influence on MRI measures of brain structure in schizophrenia. Molecular Psychiatry, 16(8), pp.826–837.
Rosa, F.W. (1991) ‘Spina bifida in infants of women treated with valproic acid’, Lancet, 338(8761), p. 125.
Rose N. (2007). The Politics of Life Itself: Biomedicine, Power, and Subjectivity in the Twenty-First Century. Princeton University Press.
Rose, N., 2019. The psychological life of neoliberalism: Neoliberalism, psychology and the birth of biopolitics. Theory, Culture & Society, 36(4), pp.1–25.
Rossignol, D. A., & frye, R.E. (2012) A review of research trends in physiologiocal abnormalities in autism spectrum disorders: immune dysregulation, inflammation and mitochondrial dysfunstion. Moleculaar Psychiatry, 17(4), 389-401.
Rossignol, D.A. and Frye, R.E. (2014). A review of research trends in physiological abnormalities in autism spectrum disorders: immune dysregulation, inflammation, oxidative stress, mitochondrial dysfunction, and environmental toxicant exposures. Molecular Psychiatry, 19(4), pp.429–439.
Rossignol, D.A. and Frye, R.E., 2014. Evidence linking oxidative stress, mitochondrial dysfunction, and inflammation in the brain of individuals with autism. Frontiers in Physiology, 5, p.150. Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3944636/
Royal Marsden NHS Foundation Trust (2024) Why are bowel cancer rates rising in younger adults? Available at: https://www.royalmarsden.nhs.uk/private-care/news-and-blogs/why-are-bowel-cancer-rates-rising-younger-adults
Rubio-Tapia, A., et al. (2009) Increased Prevalence and Mortality in Undiagnosed Celiac Disease. Gastroenterology. Available at: https://www.gastrojournal.org/article/S0016-5085(09)00878-7/fulltext
Rubio-Tapia, A., Kyle, R.A., Kaplan, E.L., Johnson, D.R., Page, W., Erdtmann, F., Brantner, T.L., Kim, W.R., Phelps, T.K., Lahr, B.D., Zinsmeister, A.R., Melton, L.J. 3rd, and Murray, J.A., 2009. Increased prevalence and mortality in undiagnosed celiac disease. Gastroenterology, 137(1), pp.88–93. https://doi.org/10.1053/j.gastro.2009.03.059
Rynecki, N.D. et al. (2023). Recent Trends in Prostate Cancer. JAMA Network Open, 6(3):e235664.
Safi, J., Joyeux, L. and Chalouhi, G.E., 2012. Periconceptional folate deficiency and implications in neural tube defects. Journal of Pregnancy, 2012, Article ID 295083. Available at: https://doi.org/10.1155/2012/295083
Sahin U, Türeci Ö. (2021). Personalized vaccines for cancer immunotherapy. Science, 359(6382), 1355–1360. doi:10.1126/science.aar7112.
Sathe, N.A. et al., 2022. Prevalence of gastrointestinal symptoms in the general population: a systematic review. BMJ Open, 12(5), pp.e056806. https://doi.org/10.1136/bmjopen-2021-056806
Satterstrom, F.K., Kosmicki, J.A., Wang, J., Breen, M.S., De Rubeis, S. and An, J.Y., et al., 2020. Large-scale exome sequencing study implicates both developmental and functional changes in the neurobiology of autism. Cell, 180(3), pp.568–584.e23. Available at: https://doi.org/10.1016/j.cell.2019.12.036 [Accessed 26 Mar. 2025]
Sauer J, Mason JB, Choi SW. Too much folate: a risk factor for cancer and cardiovascular disease? Curr Opin Clin Nutr Metab Care. 2009 Jan;12(1):30-6. doi: 10.1097/MCO.0b013e32831cec62. PMID: 19057184; PMCID: PMC2790187.
Sayers, G., Hughes, J., Feely, J., & McDonnell, R. (2009). Folic acid fortification: the Irish experience. Public Health Nutrition, 12(4), 442–445. doi:10.1017/S1368980008002611
Scaglione, F. and Panzavolta, G. (2014) ‘Folate, folic acid and 5-methyltetrahydrofolate are not the same thing’, Xenobiotica, 44(5), pp. 480–488. Available at: https://doi.org/10.3109/00498254.2013.845705 (Accessed: 1 April 2025).
Schmidt, R.J., Hansen, R.L., Hartiala, J., Allayee, H., Schmidt, L.C., Tancredi, D.J., Tassone, F. and Hertz-Picciotto, I. (2011). Prenatal vitamins, one-carbon metabolism gene variants, and risk for autism. Epidemiology, 22(4), pp.476–485.
Scientific Advisory Committee on Nutrition (2006). Folic Acid and the Prevention of Disease. The Stationery Office: London. Available at: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/339312/SACN_folic_acid_report_2006.pdf [Accessed 1 Apr. 2025].
Scientific American (2023) Autoimmunity Has Reached Epidemic Levels. We Need Urgent Action to Address It. Available at: https://www.scientificamerican.com/article/autoimmunity-has-reached-epidemic-levels-we-need-urgent-action-to-address-it/
Selhub J, Rosenberg IH. Excessive folic acid intake and relation to adverse health outcome. Biochimie. 2016;126:71–78. doi:10.1016/j.biochi.2016.04.010.
Selhub, J. (1999) ‘Homocysteine metabolism’, Annual Review of Nutrition, 19, pp. 217–246. doi:10.1146/annurev.nutr.19.1.217.
Sequeira, J.M., Piroozi, A., Abdi, S., Askarian, F. and Quadros, E.V., 2016. Prenatal and postnatal folate supplementation in a genetic mouse model of autism. Neuropharmacology, 110, pp.532–541.
Sharp, W.G. et al., 2013. Feeding problems and nutrient intake in children with autism spectrum disorders: A meta-analysis and review. Journal of Autism and Developmental Disorders, 43(9), pp.2159–2173. https://doi.org/10.1007/s10803-013-1771-5
Shu, L., Zhang, x., Zhou, j., zhu, H. and Si, C.(2023) ‘Ultra-Processed Food Consumption and Increased Risk of Metabolic Syndrome’. Frontiers Nutrition,10.doi:10.3389/fnut.2023.1211797. Available at: https://www.frontiersin.org/articles/10.3389/fnut.2023.1211797/full
Siegel, R.L., Miller, K.D. and Jemal, A. (2019). Colorectal cancer statistics, 2019. CA: A Cancer Journal for Clinicians, 69(3), pp.145–164. doi:10.3322/caac.21545.
Simko, L. and McCartney, M., 2022. Public health messaging during COVID-19: Psychological coercion, compliance, and the erosion of medical ethics. Journal of Bioethical Inquiry, 19(1), pp.45–58.
Singh, R., Garg, N., and Rao, D.N. (2023) ‘Genetic polymorphisms in folate metabolism and their impact on immunotherapeutic outcomes’, Genes and Immunity, 24(1), pp.47–59.
Smith AD, Sobczyńska-Malefora A, Green R, Reynolds EH, Refsum H. Mandatory food fortification with folic acid. Lancet Glob Health. 2022 Oct;10(10): e1395–e1396. doi:10.1016/S2214-109X(22)00378
Smith, A.D., Kim, Y.I. and Refsum, H., 2008. Is folic acid good for everyone? The American Journal of Clinical Nutrition, 87(3), pp.517–533. https://doi.org/10.1016/j.amjmed.2022.12.002 (Accessed: 1 April 2025).
Stamova, B., Green, P.G., Tian, Y., Hertz-Picciotto, I., Pessah, I.N. and Sharp, F.R., 2011. Correlations between gene expression and mercury levels in blood of boys with and without autism. Neurotoxicity Research, 19(1), pp.31–48.
Stamova, B., Sharp, F.R., and Ander, B.P. (2011). Genetic and epigenetic regulation in the developing brain and their role in neurodevelopmental disorders. Neurobiology of Disease, 41(1), pp.1–13.
Stazi, A.V., Mantovani, A., Tognoni, G. and Toccaceli, V. (2008) ‘Occurrence of neural tube defects in pregnancy: an excess of cases in a 2773-km² area in Central Italy’, International Journal of Environmental Health Research, 18(3), pp. 207–217.
Stockton, D., McKinney, P.A., McKean, M.E., Pell, J.P., MacDonald, T.M. and Brewster, D.H. (2025) ‘Trends in neural tube defects in Scotland in 2000–2021 prior to the introduction of mandatory folic acid fortification of non-wholemeal wheat flour: a population-based study’, Archives of Disease in Childhood [Preprint]. Available at: https://adc.bmj.com/content/early/2025/03/16/archdischild-2024-328295 (Accessed: 27 March 2025).
Stover, P.J. (2004) ‘Physiology of folate and vitamin B12 in health and disease’, Nutrition Reviews, 62(6 Pt 2), pp. S3–S12. Available at: https://doi.org/10.1111/j.1753-4887.2004.tb00070.x (Accessed: 1 April 2025).
Sullivan, M. L., 2020. The myth of informed consent: Coercion, trust, and pharmaceutical domination in modern medicine. Medical Humanities, 46(2), pp.113–121.
Sweeney, M.R., McPartlin, J., Weir, D.G., Daly, D. and Scott, J.M., 2007. The potential role of excessive folic acid intake in neurodevelopmental disorders: is epigenetics the key? Nutrition Reviews, 65(11), pp.504–511.
Sweeney, M.R., Staines, A., Daly, L., Traynor, A., Daly, S., Bailey, S.W., Alverson, P.B., Ayling, J.E. and Scott, J.M. (2009) ‘Persistent circulating unmetabolised folic acid in a setting of liberal voluntary folic acid fortification: Implications for further mandatory fortification?’, BMC Public Health, 9, p. 295. Available at: https://doi.org/10.1186/1471-2458-9-295 (Accessed: 1 April 2025).
TallBear K. (2013). Native American DNA: Tribal Belonging and the False Promise of Genetic Science. University of Minnesota Press.
The Independent Fetal Anticonvulsant Trust (2020). Fetal Valproate Syndrome Awareness and Advocacy. Available at: https://www.infact.org.uk (Accessed 2 Apr. 2025).
The Merck Index, 1996. An Encyclopedia of Chemicals, Drugs, and Biologicals. 12th ed. Whitehouse Station, NJ: Merck & Co., Inc.
The Times. (2024). England has Europe’s steepest rise in under-50s with bowel cancer. https://www.thetimes.co.uk/article/bowel-cancer-symptoms-diet-age-888hh9kz2
Thomas, S.L. et al. (2023) ‘Trends in pediatric cancer incidence in the United States, 2003–2019’, Cancer Epidemiology, Biomarkers & Prevention, 32(8), pp. 972–980. doi:10.1158/1055-9965.EPI-22-1325.
Thuru, X., Goossens, J. F., & Bailly, C. (2022). MR1-dependent activation of MAIT cells by folic acid photoproducts. Frontiers in Immunology, 13, 946713.
Tinker, S.C., Devine, O., Mai, C.T., Zotti, M.E., and Reefhuis, J. (2010). Estimate of the potential impact of folic acid fortification of corn masa flour on the prevention of neural tube defects. Birth Defects Research Part A: Clinical and Molecular Teratology, 88(8), pp.643–649.
Tio, M., Andrici, J., Cox, M.R. and Eslick, G.D. (2014) ‘Folate intake and the risk of prostate cancer: a systematic review and meta-analysis’, Prostate Cancer and Prostatic Diseases, 17(3), pp. 213–219. Available at: https://doi.org/10.1038/pcan.2014.17 (Accessed: 2 April 2025).
Tomita, L.Y., Almeida, V.D., Villa, L.L., Franco, E.L. and Cardoso, M.A., 2013. Polymorphisms in genes involved in folate metabolism modify the association of dietary and circulating folate and vitamin B-6 with cervical neoplasia. The Journal of Nutrition, 143(12), pp.2007–2014. doi:10.3945/jn.113.182212 Folate and Cancer – Luciana Yuki Tomita, 2016
Tomson, T., Battino, D., Bonizzoni, E., Craig, J., Lindhout, D., Sabers, A., Perucca, E. and Vajda, F. (2011) ‘Dose-dependent risk of malformations with antiepileptic drugs: an analysis of data from the EURAP epilepsy and pregnancy registry’, The Lancet Neurology, 10(7), pp. 609–617. Availableat: https://www.sciencedirect.com/science/article/abs/pii/S0890623809000422 (Accessed: 27 March 2025).
Troen, A.M., Mitchell, B., Sorensen, B., Wener, M.H., Johnston, A., Wood, B., Selhub, J., McTiernan, A., Yasui, Y. and Oral, E. (2006). Unmetabolized folic acid in plasma is associated with reduced natural killer cell cytotoxicity among postmenopausal women. The Journal of Nutrition, 136(1), pp.189–194. https://doi.org/10.1093/jn/136.1.189
U.S. Court of Appeals for the Sixth Circuit (2016) Rheinfrank v. Abbott Laboratories, No. 16-3347. Available at: https://caselaw.findlaw.com/us-6th-circuit/1849669.html (Accessed: £rd April, 2025).
U.S. Department of Justice (2012) Abbott Laboratories sentenced for misbranding drug. 7 November. Available at: https://www.justice.gov/archives/opa/pr/abbott-laboratories-sentenced-misbranding-drug (Accessed: 28 March 2025).
U.S. Department of Justice (DOJ). (2012). Abbott Laboratories to pay $1.5 billion for unlawful promotion of Depakote.
U.S. Food and Drug Administration (2010). Frances Oldham Kelsey: Medical Reviewer Who Averted a Public Health Tragedy. FDA.gov
U.S. Food and Drug Administration (FDA) (1994) ‘Withdrawal of approval of the new drug application for Paramethadione’, Federal Register, 59(138), p. 36039.
U.S. Food and Drug Administration (FDA) (2007) Correspondence with Abbott Laboratories Regarding Proposed Label Changes for Depakote. (Referenced in: Rheinfrank v. Abbott Laboratories, 2016).
U.S. Food and Drug Administration (FDA) (2013) ‘FDA Drug Safety Communication: Valproate anti-seizure products contraindicated for migraine prevention in pregnant women due to decreased IQ scores in exposed children’, FDA, 6 May. Available at: https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-valproate-anti-seizure-products-contraindicated-migraine-prevention (Accessed: 2 April 2025).
U.S. Food and Drug Administration (FDA), 2004. Drugs@FDA: FDA Approved Drug Products – Equivalence Evaluations (Orange Book). Paramethadione (Paradione) listing. Manufacturer: Abbott Laboratories.
U.S. Food and Drug Administration (FDA) (2013) Drug Efficacy Study Implementation (DESI). FDA History Office. Available at: https://www.fda.gov/about-fda/histories-product-regulation/drug-efficacy-study-implementation-desi (Accessed: 4th April, 2025).
U.S. Food and Drug Administration (2011) Drug Safety Communication: Children born to mothers who took Valproate products while pregnant may have impaired cognitive development. Available at: https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-children-born-mothers-who-took-valproate-products-while-pregnant-may (Accessed: 4th April, 2025).
U.S. Food and Drug Administration (2013) Drug Safety Communication: Valproate Anti-seizure Products Contraindicated for Migraine Prevention in Pregnant Women. Available at: https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-valproate-anti-seizure-products-contraindicated-migraine-prevention (Accessed: 4th April, 2025).
U.S. Food and Drug Administration (1996). Food standards: amendment of standards of identity for enriched grain products to require addition of folic acid. Federal Register, 61(44), pp.8781–8797.
Ueda, Y., Yamaguchi, M., Asai, Y., et al. (2016) ‘Neural tube defect prevalence in Japan after folic acid recommendations without mandatory fortification’, Congenital Anomalies, 56(1), pp. 1–7. doi:10.1111/cga.12129.
Ueland, P.M., Hustad, S., Schneede, J., Refsum, H. and Vollset, S.E. (2001) ‘Biological and clinical implications of the MTHFR C677T polymorphism’, Trends in Pharmacological Sciences, 22(4), pp. 195–201. Available at: https://doi.org/10.1016/S0165-6147(00)01675-8 (Accessed: 2 April 2025).
UK Government, 1998. Human Rights Act 1998. [online] Available at: https://www.legislation.gov.uk/ukpga/1998/42/contents [Accessed 6 Apr. 2025].
UK Government (2021) Folic acid added to flour to prevent spinal conditions in babies. Available at: https://www.gov.uk/government/news/folic-acid-added-to-flour-to-prevent-spinal-conditions-in-babies (Accessed: 3rd March, 2025).
UK Government, 2021. Government to introduce folic acid into flour to help prevent spinal conditions in babies. [online] GOV.UK. Available at: https://www.gov.uk/government/news/government-to-introduce-folic-acid-into-flour-to-help-prevent-spinal-conditions-in-babies [Accessed 31 Mar. 2025].
UK Teratology Information Service (2023) Use of high-risk medications in pregnancy. Available at: https://uktis.org/ (Accessed: 27 March 2025).
UK Teratology Information Service (2023) Use of sodium valproate in pregnancy. Available at: https://uktis.org/monographs/use-of-sodium-valproate-in-pregnancy/ (Accessed: 27 March 2025).
Ulrich, C.M. and Potter, J.D., 2006. Will mandatory folic acid fortification prevent or promote cancer? The American Journal of Clinical Nutrition, 84(1), pp.154–158. Available at: https://doi.org/10.1093/ajcn/84.1.154
Ulrich, C.M. and Potter, J.D., 2006. Will mandatory folic acid fortification prevent or promote cancer? The American Journal of Clinical Nutrition, 84(1), pp.154–158. Available at: https://doi.org/10.1093/ajcn/84.1.154
United Nations, 2006. Convention on the Rights of Persons with Disabilities (CRPD). New York: United Nations General Assembly. Available at: https://www.un.org/disabilities/documents/convention/convoptprot-e.pdf [Accessed 6 Apr. 2025].
United Nations Environment Programme (2021) Inside the 20-year campaign to rid the world of leaded fuel. Available at: https://www.unep.org/news-and-stories/story/inside-20-year-campaign-rid-world-leaded-fuel (Accessed: 27 March 2025).
United Nations Environment Programme (UNEP) (2022) ‘Children and Pregnant Mothers in Developing World Face Widespread Lead Poisoning’, UNEP Press Release. Available at: https://www.unep.org/news-and-stories/press-release/children-and-pregnant-mothers-developing-world-face-widespread (Accessed: 2 April 2025).
U.S. Congress (1962) Drug Amendments of 1962 (Kefauver-Harris Amendments). Public Law 87-781. Washington, D.C.: Government Printing Office. Available at: https://www.fda.gov/media/70985/download (Accessed: 4th April, 2025).
U.S. Senate Finance Committee, 2022. Interim Report: Big Pharma Tax Avoidance. [online] Washington D.C.: United States Senate. Available at: https://www.finance.senate.gov/imo/media/doc/Pharma%20Tax%20Report.pdf [Accessed 8 Apr. 2025].
United States District Court, Southern District of Illinois, 2017. E.R.G. v. Abbott Laboratories, Inc. (In re Depakote), Case No. 12-CV-55-NJR-SCW. [online] Available at: https://casetext.com/case/erg-v-abbott-labs-inc-in-re-depakote-2 [Accessed 22 Mar. 2025].
United States Patent and Trademark Office (USPTO). (2011). WO2012061779A1 – Pharmaceutical compositions comprising succinimide derivatives. Listed assignee: Abbott Laboratories.
Vargesson, N. (2009) ‘Thalidomide-induced limb defects: resolving a 50-year-old puzzle’, BioEssays, 31(12), pp. 1327–1336. Available at: https://pubmed.ncbi.nlm.nih.gov/19183017/ (Accessed: 27 March 2025).
Vincent, J.B. et al. (2011) ‘The role of folate receptor alpha autoantibodies in cerebral folate deficiency and neurodevelopmental disorders’, Brain Research, 1380, pp. 53–64.
Vivier, E., Ugolini, S., Blaise, D. et al. (2021) ‘Innate immunity: the role of natural killer cells in antiviral defence’, Nature Reviews Immunology, 21(6), pp. 355–368. doi:10.1038/s41577-021-00558-3.
Vollset, S. E., et al. (2013). Colorectal cancer risk and folic acid fortification. American Journal of Clinical Nutrition, 98(3), 682-687.
Von Elm, E., Altman, D.G., Egger, M., Pocock, S.J., Gøtzsche, P.C. and Vandenbroucke, J.P., 2007. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. The Lancet, 370(9596), pp.1453-1457.
Vuik, F. E. R., Nieuwenburg, S. A. V., Bardou, M., Lansdorp-Vogelaar, I., Dinis-Ribeiro, M., Bento, M. J., … & Spaander, M. C. W. (2019). Increasing incidence of colorectal cancer in young adults in Europe over the last 25 years. Gut, 68(10), 1820-1826. Available at: Increasing incidence of colorectal cancer in young adults in Europe over the last 25 years – PubMed
Wald, N.J., Law, M.R., Morris, J.K. and Wald, D.S. (2001) ‘Quantifying the effect of folic acid’, British Medical Journal, 322(7291), pp. 1399–1400. doi:10.1136/bmj.322.7291.1399.
Wallace, D.C. and Fan, W. (2010) RNA-directed DNA methylation: an epigenetic pathway of increasing complexity’, Nature Reviews Genetics, 15(6), pp. 394–408.
Wallace, dc, 2005 Energetics, epigenetics, mitochondrial genetics’, Mitochondrion, 10(1), pp. 12–31.
Walter Veit, Jonathan Anomaly, Nicholas Agar, Peter Singer, Diana S. Fleischman, Francesca Minerva (2021). “Can ‘eugenics’ be defended?” Monash Bioethics Review, 39(1), 60–67. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8321981/
Wang, Y., Wang, Z., Jiang, L., & Du, Y. (2015). Population pharmacokinetics of valproic acid in Chinese epileptic patients. Therapeutic Drug Monitoring, 37(1), 100–106. https://doi.org/10.1097/FTD.0000000000000100
Waterland, R.A. and Jirtle, R.L., 2003. Transposable elements: targets for early nutritional effects on epigenetic gene regulation. Molecular and Cellular Biology, 23(15), pp.5293–5300.
Wei, X., Chen, Y., Xu, X., Zhao, W. and Wang, X., 2008. Valproic acid affects folate metabolism in human embryonic stem cells. Toxicology Letters, 181(1), pp.19–26. doi:10.1016/j.toxlet.2008.06.862.
Weiner, L.M., et al. (1985). Fetal trimethadione and paramethadione syndrome. Teratology, 32(3), 329–336.
Weisberg, I., Tran, P., Christensen, B., Sibani, S. and Rozen, R., 1998. A second genetic polymorphism in methylenetetrahydrofolate reductase (MTHFR) associated with decreased enzyme activity. Molecular Genetics and Metabolism, 64(3), pp.169–172.
West, J., et al. (2014) Incidence and prevalence of celiac disease and dermatitis herpetiformis in the UK over two decades: population-based study. BMJ Open. Available at: https://bmjopen.bmj.com/content/4/3/e003718
Williams, L.J., Mai, C.T., Edmonds, L.D., et al. (2002) ‘Prevalence of spina bifida and anencephaly during the transition to mandatory folic acid fortification in the United States’, Teratology, 66(1), pp. 33–39. doi:10.1002/tera.10073.
Wisconsin Medical Journal Editorial Board, 2023. The folic acid debate: A reappraisal. WMJ, 123(6), pp.495–500.
World Cancer Research Fund International (2023) Reports Archive. [online] Available at: https://www.wcrf.org/resource_types/reports/ [Accessed 23 Mar. 2025].
World Cancer Research Fund International (2023) Research & Policy Library. [online] Available at: https://www.wcrf.org/research-policy/library/ [Accessed 23 Mar. 2025].
World Cancer Research Fund International (WCRF): World Cancer Research Fund International (2023) UK cancer statistics. [online] Available at: https://www.wcrf.org/preventing-cancer/cancer-statistics/uk-cancer-statistics/ (Accessed 23 Mar. 2025).
World Health Organization (2015) Guideline: Optimal serum and red blood cell folate concentrations in women of reproductive age for prevention of neural tube defects. Geneva: WHO. Available at: https://apps.who.int/iris/handle/10665/161988 (Accessed: 28 March 2025).
World Health Organization (2016) WHO Recommendations on Antenatal Care for a Positive Pregnancy Experience. Available at: https://www.who.int/publications/i/item/9789241549912 (Accessed: 27 March 2025).
World Health Organization (WHO). (2009) The influence and effects of social inequalities on environmental health risks. Copenhagen: WHO Regional Office for Europe. Available at: https://iris.who.int/bitstream/handle/10665/107965/WHO-EURO-2009-8549-48321-71741-eng.pdf (Accessed: 2 April 2025).
World Health Organization (WHO). (2022) Periconceptional folic acid supplementation to prevent neural tube defects. Geneva: World Health Organization. Available at: https://www.who.int/tools/elena/interventions/folate-periconceptional (Accessed: 2 April 2025).
World Health Organization (WHO). (2022). Neural tube defects and prevention strategies: Global review.
World Health Organization (2002) The Importance of Pharmacovigilance: Safety Monitoring of Medicinal Products. Geneva: WHO. Available at: https://apps.who.int/iris/handle/10665/42493 (Accessed: [insert date]).
World Health Organization (WHO). (2025) Mapping the commercial determinants of health in countries of the South-East Asia Region. New Delhi: WHO Regional Office for South-East Asia. Available at: https://iris.who.int/bitstream/handle/10665/380673/9789290220336-eng.pdf (Accessed: 2 April 2025).
Wozniak, R.H. and Bianchi, F. (2019) ‘Missing metrics: data suppression in neural tube defect surveillance post-fortification’, Global Health Review, 15(2), pp. 47–56.
Wright, A.J.A., Dainty, J.R. and Finglas, P.M. (2007) ‘Folic acid metabolism in human subjects revisited: potential implications for proposed mandatory folic acid fortification in the UK’, British Journal of Nutrition, 98(4), pp. 667–675. Available at: https://doi.org/10.1017/S0007114507746896 (Accessed: 2 April 2025).
Xu, M., Zhang, J. and Wu, Z. (2024) ‘Impacts of food additives on gut microbiota and host health’, Food Research International, 179, Article 113888. Available at: https://www.sciencedirect.com/science/article/abs/pii/S0963996924010688 (Accessed: 2 April 2025).
Yadav, D.K., Tiwari, A., and Singh, V. (2023) ‘MTHFR polymorphism and its potential influence on COVID-19 vaccine response and complications: A mechanistic overview’, Clinical Genetics and Genomics, 5(1), pp.1–10.
Yin, S.A. et al. (2012) ‘Effect of maternal folic acid supplement dosage on neural tube defects and other pregnancy outcomes in northern China: A large prospective cohort study’, BMJ Open, 2(4), e001800.
Yin, T., Zeng, Y., Zhang, X., Wang, W., Ren, X. and Zhang, X. (2012) ‘Aberrant placenta gene expression pattern in bovine pregnancies established with cloned embryos’, Physiological Genomics, 44(12), pp. 607–616. Available at: https://doi.org/10.1152/physiolgenomics.00076.2012 (Accessed: 2 April 2025).
Zackai, E.H., Mellman, W.J., Neiderer, B. and Hanson, J.W. (1975) ‘The fetal trimethadione syndrome’, The Journal of Pediatrics, 87(2), pp. 280–284. Available at: https://doi.org/10.1016/S0022-3476(75)80603-2 (Accessed: 2 April 2025).
Zeisel, S.H. (2009) ‘Epigenetic mechanisms for nutrition determinants of later health outcomes’, The American Journal of Clinical Nutrition, 89(5), pp. 1488S–1493S. Available at: https://doi.org/10.3945/ajcn.2008.26834C (Accessed: 2 April 2025).
ZERO Prostate Cancer (2024) Prostate cancer facts and statistics. [online] Available at: https://zerocancer.org/about-prostate-cancer/facts-statistics [Accessed 23 Mar. 2025].
Zhang, X., Platt, R.W. and Cnattingius, S. (2007) ‘Reduced birthweight in infants after maternal periconceptional exposure to tobacco smoke’, Paediatric and Perinatal Epidemiology, 21(Suppl 1), pp. 35–45. Available at: https://doi.org/10.1111/j.1365-3016.2007.00861.x (Accessed: 2 April 2025).
Zhao, E., Maj, T., Kryczek, I., Li, W., Wu, K., Zhao, L., Wei, S., Crespo, J., Wan, S., Vatan, L., et al. (2023) ‘Epigenetic regulation of tumor-infiltrating T cells’, Nature Immunology, 24(1), pp. 120–130. Available at: https://doi.org/10.1038/s41590-022-01365-3 (Accessed: 2 April 2025).
Zhao, J. et al. (2019) ‘High-dose folic acid supplementation and risk of gestational hypertension’, Hypertension, 74(1), pp. 130–137. doi:10.1161/HYPERTENSIONAHA.119.14621.
Zhao, Y., Xu, Y., Wang, Y. et al. (2024) ‘Natural killer cells in tumour immunosurveillance and therapy’, Cell Death & Disease, 15(1), Article 45. doi:10.1038/s41419-024-06976-0.
Zhou, Y., Han, T., Chen, J., et al. (2023). The risk of new-onset autoimmune diseases following COVID-19: a systematic review and meta-analysis. Frontiers in Immunology, 14, 1113170.
Zsigra, S., et al. (2022). Folic Acid Treatment Directly Influences the Genetic and Epigenetic Regulation along with the Associated Cellular Maintenance Processes of HT-29 and SW480 Colorectal Cancer Cell Lines. Cancers (Basel), 14(7), 1820. Available at: https://doi.org/10.3390/cancers14071820
ADDENDUM: 24/04/2025
It is important to note that the original source for this citation -Johns Hopkins Bloomberg School of Public Health (2016), “Too Much Folate in Pregnant Women Increases Risk for Autism” -was initially published under the URL:
However, this link later returned a 404 error and appeared inaccessible, creating the impression that the source had been removed. It was subsequently discovered that the page had been silently relocated to a revised URL, with the trailing word “study” removed, now available at:
This kind of unannounced link breakage without proper redirect protocols (i.e. lack of 301 or 302 HTTP redirects) undermines research integrity and obscures the audit trail for verifiable sources. As noted by Berners-Lee et al. (2013), the persistence of URLs is essential for maintaining scholarly transparency and citation continuity. The failure to implement redirects or metadata indexing in institutional websites effectively discredits independent researchers who cite sources in good faith, only to have those sources appear invalid after backend changes.
This incident mirrors a growing pattern of “soft suppression” wherein controversial or inconvenient studies are not deleted, but are made difficult to trace, cite, or defend -especially in real-time investigations. The digital infrastructure exists to prevent this (via canonical URLs, archive embedding, and dynamic redirects), yet institutions routinely fail to apply it when politically sensitive material is involved (Klein, 2018).
To preserve record integrity, researchers are advised to archive all source pages upon access (via the Internet Archive or archive.today), store local PDF copies, and document any changes in URL pathing. This case is one such example of silent information drift—a tactic that must be acknowledged and resisted within scholarly discourse.
References
Berners-Lee, T., Hall, W., Hendler, J., O’Hara, K., Shadbolt, N. and Weitzner, D., 2013. A Framework for Web Science. Foundations and Trends in Web Science, 1(1), pp.1–130.
Klein, M., 2018. Scholarly Context Not Found: One in Five Articles Suffers from Reference Rot. PLOS ONE, [online] 9(12). Available at: https://doi.org/10.1371/journal.pone.0115253.
